Abstract
The biosynthesis and processing of wheat-germ agglutinin (WGA) were studied in developing wheat (Triticum aestivum L. cv. Marshall) embryos using pulse-chase labeling, subcellular fractionation and immunocytochemistry. A substantial amount of newly synthesized WGA was organelle-associated. Isolation of WGA on affinity columns of immobilized N-acetylglucosamine indicated that it was present in a dimeric form. When extracts from embryos pulse-labeled with [35S]cysteine were fractionated on an isopycnic sucrose gradient, radioactivity incorporated into WGA was detected at a position coincident with the endoplasmic reticulum (ER) marker enzyme NADH-cytochromec reductase. The WGA in the ER could be slowly chased into the soluble, vacuolar fraction, with a half-life of approx. 8 h. Immunolocalization studies demonstrated the accumulation and distribution of WGA throughout the vacuoles.
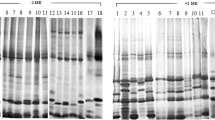
Four forms of the WGA monomer were characterized using immunoaffinity purification and sodium dodecyl sulfate-polyacrylamide gel electrophoresis. In-vitro translation of polyadenylated RNA isolated from developing wheat embryos produced a polypeptide with Mr 21 000. In-vivo labeling of embryos with radioactive amino acids resulted in the formation of a polypeptide of Mr 23 000 and the mature monomer of Mr 18000. When [3H]mannose was used in labeling studies, only the polypeptide of Mr 23 000 was detected. In-vivo labeling in the presence of tunicamycin yielded an additional polypeptide of Mr 20 000. These results indicate that WGA is cotranslationally processed by the removal of a signal peptide and the addition of a glycan, presumably at the carboxy-terminus (N.V. Raikhel and T.A. Wilkins, 1987, Proc. Natl. Acad. Sci. USA 84, 6745–6749). The glycosylated precursor of WGA is post-translationally processed to the mature form by the removal of a carboxyl-terminal glycopeptide.
Similar content being viewed by others
Abbreviations
- ER:
-
endoplasmic reticulum
- IgG:
-
immunoglobulin G
- Mr :
-
relative molecular mass
- poly(A)+RNA:
-
polyadenylated RNA
- SDS-PAGE:
-
sodium dodecyl sulfatepolyacrylamide gel electrophoresis
- WGA:
-
wheat-germ agglutinin
References
Baulcombe, D.C., Key, J.L. (1980) Polyadenylated RNA sequences which are reduced in concentration following auxin treatment of soybean hypocotyls. J. Biol. Chem.255, 8907–8913
Bishop, J.O., Rosbash, M., Evans, D. (1974) Polynucleotide sequences in eukaryotic DNA and RNA that form ribonuclease-resistant complexes with polyuridylic acid. J. Mol. Biol.85, 75–86
Bollini, R., Vitale, A., Chrispeels, M.J. (1983) In vivo and in vitro processing of seed reserve protein in the endoplasmic reticulum: evidence for two glycosylation steps. J. Cell Biol.96, 999–1007
Bowles, D.J., Marcus, S.E., Pappin, D.J.C., Findlay, J.B.C., Eliopoulos, E., Maycox, P.R., Burgess, J. (1986) Posttranslational processing of concanavalin A precursors in jackbean cotyledons. J. Cell Biol.102, 1284–1297
Chrispeels, M.J. (1984) Biosynthesis, processing and transport of storage proteins and lectins in cotyledons of developing legume seeds. Phil. Trans. R. Soc. London Ser. B304, 309–322
Chrispeels, M.J., Hartl, P.M., Sturm, A., Faye, L. (1986) Characterization of the endoplasmic reticulum-associated precursor of concanavalin A. J. Biol. Chem.261, 10021–10024
Chrispeels, M.J., Higgins, T.V.J., Craig, S., Spencer, D. (1982) Role of the endoplasmic reticulum in the synthesis of reserve proteins and the kinetics of their transport to protein bodies in developing pea cotyledons. J. Cell Biol.93, 5–14
Craig, S., Goodchild, D.J. (1984) Periodate-acid treatment of sections permits on-grid immunogold localization of pea seed vicilin in ER and Golgi. Protoplasma122, 35–44
Crouch, M.L., Tenbarge, K.M., Simon, A.E., Ferl, R. (1983) cDNA clones forBrassica napus seed storage proteins: evidence from nucleotide sequence analysis that both subunits of napin are cleaved from a precursor polypeptide. J. Mol. Appl. Gen.2, 273–283
Ericson, M.L., Rodin, J., Lenman, L., Glimelius, K., Josefsson, L.G., Rask, L. (1986) Structure of the rapeseed 1.7 S storage protein, napin, and its precursor. J. Biol. Chem.261, 14576–14581
Faye, L., Chrispeels, M.J. (1987) Transport and processing of the glycosylated precursor of concanavalin A in jack-bean. Planta170, 217–224
Herman, E.M., Shannon, L.M., Chrispeels, M.J. (1985) Concanvalin A is synthesized as a glycoprotein precursor. Planta165, 23–29
Higgins, T.J.V. (1984) Synthesis and regulation of major proteins in seeds. Annu. Rev. Plant Physiol.35, 191–221
Higgins, T.J.V., Chandler, P.M., Zurawski, G., Button, S.C., Spencer, D. (1983) The biosynthesis and primary structure of pea seed lectin. J. Biol. Chem.258, 9544–9549
Hubbard, S.C., Ivatt, R.J. (1981) Synthesis and processing of asparagine-linked oligosaccharides. Annu. Rev. Biochem.50, 555–583
Jackson, M., Ingle, J. (1973) The interpretation of studies on rapidly labeled ribonucleic acid in higher plants. Plant Physiol.51, 412–414
Laemmli, U.K. (1970) cleavage of structural proteins during the assembly of the head of bacteriophage T-4. Nature227, 680–685
Lord, J.M. (1985) Precursor of ricin andRicinus communis agglutinin. Glycosylation and processing during synthesis and intracellular transport. Eur. J. Biochem.146, 411–416
Mishkind, M.L., Keegstra, K., Palevitz, B.A. (1980) Distribution of wheat germ agglutinin in young wheat plants. Plant Physiol.66, 950–955
Mishkind, M.L., Raikhel, N.V., Palevitz, B.A., Keegstra, K. (1982) Immunocytochemical localization of wheat germ agglutinin in wheat. J. Cell Biol.92, 753–764
Mishkind, M.L., Raikhel, N.V., Palevitz, B.A., Keegstra, K. (1983) The cell biology of wheat germ agglutinin and related lectins. In: Chemical taxonomy, molecular biology, and function of plant lectins, pp. 163–176, Goldstein, I.J., Etzler, M.E., eds. Alan R. Liss, New York
Nagata, Y., Burger, M.M. (1974) Wheat germ agglutinin. J. Biol. Chem.149, 3116–3122
Quatrano, R.S., Hopkins, R., Raikhel, N.V. (1983) Control of the synthesis and localization of wheat germ agglutinin during embryogenesis. In: Chemical taxonomy, molecular biology, and function of plant lectins, pp. 117–130, Goldstein, I.J., Etzler, M.E., eds. Alan R. Liss, New York
Raikhel, N.V., Mishkind, M.L., Palevitz, B.A. (1984a) Characterization of a wheat germ agglutinin-like lectin from adult wheat plants. Planta162, 55–61
Raikhel, N.V., Mishkind, M.L., Palevitz, B.A. (1984b) Immunocytochemistry in plants with colloidal gold conjugates. Protoplasma121, 25–33
Raikhel, N.V., Quatrano, R.S. (1986) Localization of wheatgerm agglutinin in developing wheat embryos and those cultured in abscisic acid. Planta168, 433–440
Raikhel, N.V., Wilkins, T.A. (1987) Isolation and characterization of a cDNA clone encoding wheat germ agglutinin. Proc. Natl. Acad. Sci. USA84, 6745–6749
Silflow, C.D., Hammett, J.R., Key, J.L. (1979) Sequence complexity of polyadenylated ribonucleic acid from soybean suspension culture cells. Biochem.18, 2725–2731
Stinissen, H., Peumans, W.J., Carlier, A. (1982) In vivo synthesis and processing of cereal lectins. Plant Mol. Biol.1, 277–290
Stinissen, H., Peumans, W.J., Chrispeels, M.J. (1984) Subcellular site of lectin synthesis in developing rice embryos. EMBO J.3, 1979–1985
Sun, S.S.M., Altenbach, S.B., Leung, F.W. (1987) Properties, biosynthesis and processing of a sulfur-rich protein in Brazil nut (Bertholletia excelsa H.B.K.). Eur. J. Biochem.162, 477–483
Triplett, B.A., Quatrano, R.S. (1982) Timing, localization, and control of wheat germ agglutinin synthesis in developing wheat embryos. Dev. Biol.91, 491–496
Wright, C.S. (1987) Refinement of the crystal structure of wheat germ agglutinin isolectin 2 at 1.8 A resolution. J. Mol. Biol.194, 501–529
Wright, C.S., Olafsdittir, S. (1986) Structural differences in the two major wheat germ agglutinin isolectins. J. Biol. Chem.261, 7191–7195
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Mansfield, M.A., Peumans, W.J. & Raikhel, N.V. Wheat-germ agglutinin is synthesized as a glycosylated precursor. Planta 173, 482–489 (1988). https://doi.org/10.1007/BF00958961
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00958961