Conclusions
-
1.
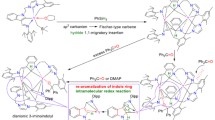
Two types of isomeric chelate compounds have been prepared in which deprotonated N-organyl-N′-(1,3-thiazol-2-yl)thiourea coordinates with R2B at the N atom of the thiazole ring and at the S or N atom of the thiocarbamide group (S- and N-isomers), the N-isomers being thermodynamically more stable than the S-isomers.
-
2.
Complexes of diorganylboryl[2-(N-organyl)carbamoylaminothiazolates] (N-isomers) with Ni and Pd have been prepared.
Similar content being viewed by others
Literature cited
V. A. Dorokhov, O. G. Boldyreva, B. M. Mikhailov, et al., Izv. Akad. Nauk SSSR, Ser. Khim., 431 (1985).
V. A. Dorokhov and O. G. Boldyreva, Advances in the Chemistry of Nitrogen Heterocycles [in Russian], Rostov-on-Don (1983), p. 38.
V. A. Dorokhov, L. I. Lavrinovich, M. N. Bochkareva, et al., Izv. Akad. Nauk SSSR, Ser. Khim., 1340 (1979).
T. Tatantelli and C. Furlani, J. Chem. Soc., A, 1217 (1971).
B. M. Mikhailov, V. A. Dorokhov, and V. I. Seredenko, Izv. Akad. Nauk SSSR, Ser. Khim., 1385 (1978).
Houben-Weil, Methoden der Organischen Chemie, Vol. 8, Georg Thieme Verlag, Stuttgart (1952), p. 157.
Author information
Authors and Affiliations
Additional information
Translated from Izvestiya Akademii Nauk SSSR, Seriya Khimicheskaya, No. 12, pp. 2822–2825, December, 1987.
Rights and permissions
About this article
Cite this article
Dorokhov, V.A., Boldyreva, O.G. & Bochkareva, M.N. Chelates of boron with N-organyl-N′-(1,3-thiazol-2-YL)thioureas and their use as ligands for complexes with transition metals. Russ Chem Bull 36, 2619–2622 (1987). https://doi.org/10.1007/BF00957248
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00957248