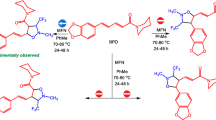
Conclusions
-
1.
Phenyl radicals add to the phenyl esters of thiocarboxylic acids
(R=Ph, Ph2CH, PhCH2) at the sulfur atom of the C=S group with the formation of radical-adducts of the type
, which were identified by ESR using a spin trap technique.
-
2.
The radical-adduct Ph2
is converted into the O,S-bis-phenylacetal of diphenylketene and the phenyl ester of diphenylthioacetic acid while the radical-adduct
is converted into the phenyl ester of thiobenzoic acid and diphenyldisulfide.
Similar content being viewed by others
Literature cited
R. G. Gasanov, R. G. Petrova, I. O. Bragina, and R. Kh. Freidlina, Izv. Akad. Nauk SSSR, Ser, Khim., 2464 (1977).
R. G. Petrova, R. G. Gasanov, and T. D. Churkina, Izv. Akad. Nauk SSSR, Ser. Khim., 379 (1981).
R. Kh. Freidlina, I. I. Kandror, and B. V. Kopyiova, Izv. Akad. Nauk SSSR, Ser. Khim., 1799 (1983).
D. H. Barton and S. M. Combil, J. Chem. Soc., Perkin Trans, 1, 1574 (1975).
O. Kajimoto, M. Kobayashi, and T. Fueno, Bull. Chem Soc. Jpn.,46, 1422 (1973).
V. M. Bzhezovskii, G. A. Kalabin, I. A. Aliev, B. A. Trofimov, M. A. Shakhgel'diev, and M. M. Kuilev, Izv. Akad. Nauk SSSR, Ser. Khim., 1999 (1976).
B. S. Perdersen, S. Scheibye, K. Clausen, and S.-O. Lawesson, Bull. Chem. Soc. Belg.,87, 293 (1978).
B. S. Perdersen, S. Schiebye, N. H. Nilson, and S.-O. Lawesson, Bull. Chem. Soc. Belg.,87, 227 (1978).
J. Voss and K. Schlapkohl, Tetrahedron,31, 2982 (1975).
H. France, I. M. Heilbron, and D. H. Hey, J. Chem. Soc., 369 (1940).
Author information
Authors and Affiliations
Additional information
Translated from Izvestiya Akademii Nauk SSSR, Seriya Khimicheskaya, pp. 1576–1581, July, 1984.
Rights and permissions
About this article
Cite this article
Petrova, R.G., Gasanov, R.G., Churkina, T.D. et al. Investigation of the reaction of phenyl radicals with phenyl esters of thiocabboxylic acids. Russ Chem Bull 33, 1447–1451 (1984). https://doi.org/10.1007/BF00956523
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00956523