Conclusions
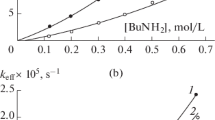
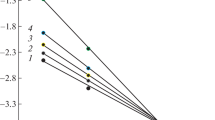
The relationship between the free energies of activation ΔG≠ and reaction ΔGo for proton transfer processes have been analyzed, taking into account the effect of hindered rotation of the reagents. We have shown that the considered effect can considerably affect the shape of the ΔG≠=f(ΔGo) curve.
Similar content being viewed by others
Literature cited
R. R. Dogonadze and A. M. Kuznetsov, Results of Science and Technology: Physical Chemistry. Kinetics [in Russian], Vol. 2, VINITI, Moscow (1973).
A. M. Kuznetsov, Elektrokhimiya,22, 240 (1986).
A. M. Kuznetsov, Élektrokhimiya,22, 291 (1986).
É. D. German and A. M. Kuznetsov, Results of Sciences and Technology: Kinetics and Catalysis [in Russian], VINITI, Moscow (1982), Vol. 10, p. 115.
N. G. Zharova, I. O. Shapiro, I. A. Misurkin, and É. D. German, Izv. Akad. Nauk SSSR, Ser. Khim., No. 2 (1989).
H. Eyring, J. Walter, and G. E. Kimball, Quantum Chemistry, Wiley, New York (1944).
I. O. Shapiro, Physical Chemistry, in: Contemporary Problems (Ya. M. Kolotyrkin, ed.) [in Russian], Khimiya, Moscow (1987), p. 128.
Author information
Authors and Affiliations
Additional information
Translated from Izvestiya Akademii Nauk SSSR, Seriya Khimicheskaya, No. 1, pp. 77–81, January 1989.
Rights and permissions
About this article
Cite this article
German, É.D., Kuznetsov, A.M. Effect of intramolecular rotational reorganization of reagents on the ratio between free energies of activation and reaction. Russ Chem Bull 38, 66–69 (1989). https://doi.org/10.1007/BF00953702
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00953702