Conclusions
-
1.
The kinetics of the thermal decomposition of dimethyltriazanium (DMTA) chloride and nitrate is described by a first-order autocatalysis equation.
-
2.
The major contribution to autoacceleration of decomposition is made by the transfer of salts to the liquid phase as a result of their solubility in their own decomposition products.
-
3.
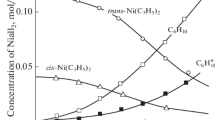
The decomposition of DMTA salts proceeds by the elimination of an NH2 + cation and methane from the [(CH3)2N(NH2)2]+ cation.
Similar content being viewed by others
Literature cited
G. B. Manelis, V. K. Strunin, and Yu. I. Rubtsov, Archiwum Proceson Spalania,6, No. 1, 51–58 (1975).
Yu. L. Rubtsov, L. P. Andrienko, A. P. D'yakov, V. A. Strunin, E. N. Loginova, K. V. Titova, and V. Ya. Rosoloyskii, in: Chemical Physics of Combustion and Explosion Processes. Chemical Reaction Kinetics [in Russian], Chernogolovka (1980), pp. 91–94.
R. Gösl, Angewandte Chem., Int. Ed., No. 1, 405 (1962).
K. H. Linke, R. Turley, and W. Gössel, Z. Naturforsch.,278, 1005–1006 (1972).
Yu. N. Matyushin, T. S. Kon'kova, K. V. Titova, E. N. Loginova, A. B. Vorob'ev, and Yu. A. Lebedev, Izv. Akad. Nauk SSSR, Ser. Khim., 2392 (1981).
Yu. N. Matyushin, T. S. Kon'kova, A. B. Vorob'ev, E. N. Loginova, K. V. Titova, and Yu. A. Lebedev, Izv. Akad. Nauk SSSR, Ser. Khim., 1735 (1981).
Yu. I. Rubtsov and G. B. Manelis, Zh. Fiz. Khim.,46, 627 (1972).
V. A. Koroban, V. M. Chugunkin, and V. I. Loboda, Combustion and Explosion. Proceedings of the Third All-Union Symposium on Combustion and Explosion [in Russian], Nauka, Moscow (1972), p. 775.
Yu. I. Rubtsov and G. B. Manelis, Zh. Fiz. Khim., Collection: Chemical Kinetics and Catalysis [in Russian], Nauka, Moscow (1979), p. 98.
Author information
Authors and Affiliations
Additional information
Translated from Izvestiya Akademii Nauk SSSR, Seriya Khimicheskaya, No. 9, pp. 1953–1957, September, 1982.
Rights and permissions
About this article
Cite this article
Rubtsov, Y.I., Andrienko, L.P., Titova, K.V. et al. Kinetics of the thermal decomposition of dimethyltriazanium chloride and nitrate. Russ Chem Bull 31, 1725–1729 (1982). https://doi.org/10.1007/BF00952363
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00952363