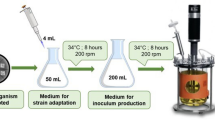
Summary
Fifty-five organisms comprising 40 bacteria, eleven yeasts and four moulds were isolated from various habitats and tested for the production of the enzyme linamarase using the release of HCN and glucose from linamarin. Only seven organisms were shown to produce linamarase: the bacteriaLeuconostoc mesenteroides, Alcaligenes faecalis, the yeastsSaccharomyces cerevisiae andRhodotorula minuta, and the mouldsAspergillus flavus, A. niger andFusarium oxysporum. A preliminary screening method used the ability to break down the glucosidep-nitrophenyl--D glucoside (PNPG). Organisms breaking down PNPG also broke down linamarin and vice-versa. The highest linamarase producers among the yeasts and bacteria as determined by HCN liberation from cassava pulp were the yeastsSacch. cerevisiae andR. minuta in about 80 and 96 hours, respectively. SinceSaccharomyces sp. withstood the highest concentration of the cyanide ion it is suggested as the organism of preference among the isolates for cassava pulp detoxication. Storage of linamarase-producing organisms in refrigerated cassava pulp was found to be a suitable method of preserving and transporting them.
Résumé
Cinquante-cinq organismes comprenant 40 bactéries, 11 levures et 4 moisissures ont été isolés à partir de différents habitats et testés pour la production de l'enzyme linamarase qui utilise le HCN et le glucose provenant de la linamarine. Une méthode de criblage préliminaire utilise l'aptitude à dégrader lep-nitrophényl-D-glucoside (PNPG). Les organismes qui dégradent le PNPG dégradent aussi la linamarine et réciproquement. Sept organismes seulement produisent la linamarase: les bactériesLeuconostoc mesenteroides etAlcaligenes faecalis, les levuresSaccharomyces cerevisiae etRhodotorula minuta, et les moisissuresAspergillus flavus, A. niger etFusarium oxysporum. D'après la vitesse de libération d'HCN à partir de la pulpe de manioc, les meilleurs producteurs de linamarase sont les levuresS. cerevisiae etR. minuta agissant, respectivement, en 80 et 96 heures environ.Saccharomyces étant parmi les organismes isolés celui qui tolère la concentration en ion cyanure la plus élevée, son utilisation est proposée pour détoxifier la pulpe de manioc. La conservation des organismes producteurs de linamarase dans la pulpe de manioc réfrigérée est la méthode proposée pour la préserrvation et le transport de ces organismes.
Resumen
Cincuenta y cinco organismos, incluyendo cuarenta bacterias, once levaduras y cuatro hongos se aislaron a partir de distintos hábitats y se estudió su producción de enzyma linamarasa, determinada por la liberación de HCN y glucosa a partir de linamarina. Tan solo siete organismos producían linamarasa: las bacteriasLeuconostoc mesenteroides yAlcaligenes faecalis, las levadurasSaccharomyces cerevisiae yRhodotorula minuta y los hongosAspergillus flavus, A. niger yFusarium oxysporum. Como método de estudio preliminar se utilizó la habilidad para degradar el glucósidop-nitrofenil-l-glucósido (PNPG), ya que los organismos capaces de degradar PNPG también eran capaces de degradar linamarina y viceversa. Entre bacterias y levaduras los mayores productores de linamarasa, expresada como cantidad de HCN liberado a partir de pulpa de casava, fueronSaccharomyces cerevisiae yRhodotorula minuta, a las 80 y 96 horas respectivamente. Al soportarSaccharomyces sp. la mayor de las concentraciones de HCN ensayadas se ha sugerido que este podría ser considerado, de entre los organismos aislados, como el más idoneo para ser utilizado en la detoxificación de la pulpa de casava. Se considera que un método adecuado para la preservación de organismos productores de linamarasa es su almacenamiento en pulpa de casava refrigerada.
Similar content being viewed by others
References
Abe, M. O. &Lindsay, R. C. 1978 Evidence for a lactic streptococcal role in Nigeria acidic cassava (Manihot esculenta Crantz) fermentations.Journal of Food Protection 41, 781–784.
Akinrele, I. A. 1964 Fermentation of cassava.Journal of the Science of Food and Agriculture 15, 589–594.
Alexopoulos, C. J. 1962Introductory Mycology 2nd edn. pp. 222–227, 228. New York: John Wiley & Sons.
Buchanan, R. E. &Gibbons, N. C. 1974Bergey's Manual of Determinative Bacteriology 8th edn. Baltimore: The Williams and Wilkins Co.
Collard, P. &Levi, S. A. 1959 A two-stage fermentation of cassava.Nature, London 183, 620–621.
Collins, C H. &Lyne, P. M. 1976Microbiological Methods 4th edn. London: Butterworths.
Cooke, R. D. 1978 An enzymatic assay for the total cyanide content of cassava (Manihot esculenta Crantz)Journal of the Science of Food and Agriculture 345–352.
Cooke, R. D. &Coursey, D. G. 1981 Cassava: a major cyanide containing food crop. InCyanide in Biology, ed. Vennesland, B., Conn, E. E., Knowles, C. J., Westley, J. & Wissing, F. pp. 93–114. New York: Academic Press.
Cowan, S. T. &Steel, K. J. 1970Manual for the Identification of Medical Bacteria, pp. 30–50, and 75–82. London: Cambridge University Press.
Davis, R. H. 1981 Cyanide detoxication in the domestic fowl. InCyanide in Biology, ed. Vennesland, B., Conn, E. E., Knowles, C. J., Westley, J. & Wissing, F. pp. 51–60. New York: Academic Press.
Ejiofor, M. A. N. 1984 Studies on microbial hydrolysis of linamarin in fermenting cassava (Manihot esculenta Crantz) pulp. Ph.D. thesis, University of Nigeria, Nsukka.
Ejiofor, M. A. N. &Okafor, N. 1981 Comparison of pressed and unpressed cassava pulp for garri making. InTropical Root Crops: Research Strategies for the 1980's, ed. Terry, E. R., Oduro, K. A. & Cavenness, F. pp. 154–158. Ottawa, Canada: I.D.R.C.
Ekpechi, O. L. 1959 Endemic goitre and high cassava diets in Eastern Nigeria. InChronic Cassava Toxicity, ed. Nestel, B. & MacIntyre, R. pp. 139–145. Ottawa, Canada: I.D.R.C.
Ikediobi, C. O., Onyia, G. O. C. &Eluwa, C. E. 1980 A rapid and inexpensive enzymatic assay for total cyanide in cassava (Manihot esculenta Crantz) and cassava products.Agricultural and Biological Chemistry 44, 2803–2808.
Jorgensen, K. 1955 Microestimation of cyanide with a modified Epstein procedure.Acta chemica scandinavica 9, 548.
Nartey, F. 1981 Cyanogenesis in tropical feeds and foodstuffs. InCyanide in Biology, ed. Vennesland, B., Conn, E. E., Knowles, C. J., Westley, J. & Wissing, F. pp. 115–132. New York: Academic Press.
Ngaba, R. R. &Lees, J. S. 1979 Fermentation of cassava (Manihot esculenta Crantz).Journal of Food Science 144, 1570–1571.
Okafor, N. 1965 Micro-organisms associated with dead insect larvae in Nigeria.Nature, London 208, 1015–1016.
Okafor, N. 1977 Micro-organisms associated with cassava fermentation for garri production.Journal of Applied Bacteriology 41, 279–284.
Okafor, N. &Ejiofor, M. A. N. 1985 The linamarase ofLeuconostoc mesenteroides: production, isolation and some properties.Journal of the Science of Food and Agriculture 36, (in press).
Okafor, N., Ijioma, B. &Oyolu, C. 1984 Studies on the microbiology of cassava retting for foo-foo production.Journal of Applied Bacteriology 56, 1–13.
Oke, O. L. 1968 Cassava as food in Nigeria.World Reviews of Nutrition and Dietetics 9, 227–250.
Osuntokun, B. O. 1970 A degenerative neuropathy with blindness and chronic cyanide intoxication of dietary origins. The evidence in Nigerians. InToxicity in the Tropics, ed. Smith, S. L. & Bababunmi, E. A. pp. 16–52. London: Taylor and Francis Ltd.
Seigler, D. S. 1981 Cyanogenic glucosides and lipids: structural types and distribution. InCyanide in Biology, ed. Vennesland, B., Conn, E. E., Knowles, C. J., Westley, J. & Wissing, F. pp. 133–143. New York: Academic Press.
Skerman, V. B. D. 1967A Guide to the Identification of Genera of Bacteria, pp. 22–31. Baltimore: Williams and Williams Co.
Smith, G. 1971An Introduction to Industrial Mycology. 6th edn. pp. 59–63. London: Edward Arnold (Publishers) Ltd.
Wood, T. 1965 The isolation, properties and enzymatic breakdown of linamarin from cassava.Journal of the Science of Food and Agriculture 16, 300–305.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Okafor, N., Ejiofor, M.A.N. The microbial breakdown of linamarin in fermenting pulp of cassava (Manihot esculenta Crantz). Mircen Journal 2, 327–338 (1986). https://doi.org/10.1007/BF00933499
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00933499