Conclusions
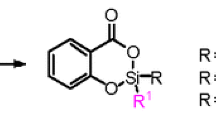
The 1,3-anionic rearrangement of aryloxysilanes is an intramolecular process that does not depend on the nature of the migrating organosilyl group but is controlled by the reactivities of the labile halogen of the aryloxysilane and of the organolithium compound in the halogen-lithium exchange reaction, which generates the silyloxyarenide ion.
Similar content being viewed by others
Literature cited
D. V. Muslin, G. A. Razuvaev, N. N. Vavilina, and N. S. Vasileiskaya, Izv. Akad. Nauk SSSR, Ser. Khim.,1975, 182.
G. A. Razuvaev, D. V. Muslin, N. S. Vasileiskaya, and F. B. Gershanov, Proceedings of the Fourth International Symposium on the Chemistry of Organosilicon Compounds, Vol. 1, Part 2, p. 160 (1975).
R. West, R. Lowe, H. F. Stewart, and A. Wright, J. Am. Chem. Soc.,93, 282 (1971).
W. J. Trepka and R. J. Sonnenfeld, J. Organomet. Chem.,16, 317 (1969).
D. E. Applequist and D. F. O'Brien, J. Am. Chem. Soc.,85, 743 (1963).
J. L. Speier, J. Am. Chem. Soc.,74, 1003 (1952).
G. D. Cooper, J. Org. Chem.,26, 925 (1961).
G. A. Razuvaev, N. S. Vasileiskaya, D. V. Muslin, N. N. Vavilina, and S. N. Uspenskaya, Zh. Org. Khim.,6, 980 (1970).
Author information
Authors and Affiliations
Additional information
Translated from Izvestiya Akademii Nauk SSSR, Seriya Khimicheskaya, No. 4, pp. 890–893, April, 1978.
Rights and permissions
About this article
Cite this article
Muslin, D.V., Lyapina, N.S., Kut'in, A.P. et al. Reactions of haloaryloxysilanes with organolithium compounds. Russ Chem Bull 27, 770–773 (1978). https://doi.org/10.1007/BF00925304
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00925304