Conclusions
-
1.
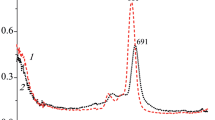
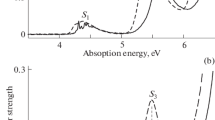
In o-chloromercuryphenols there is no intramolecular hydrogen bond with the HgCl group, while the OH group is primarily in the trans-position with respect to it.
-
2.
An increase in the ability for autoassociation of o-chloromercury derivatives of phenol was established. The ability to form hydrogen bonds with coordinating solvents was evaluated quantitatively.
Similar content being viewed by others
Literature cited
B. G. Gowenlock and J. Trotman, J. Chem. Soc., 1454 (1955).
V. Baliah and P. Subbarayan, J. Indian Chem. Soc.,40, 638 (1963).
G. Leandri and A. Tundo, J. Chem. Soc., 3377 (1954).
L. Bellamy, New Data on the IR Spectra of Complex Molecules [Russian translation], Mir (1971), p. 112.
E. Asmus, Z. Analyt. Chem.,178, 104 (1969).
M. T. Tribble and J. C. Traynham, J. Amer. Chem. Soc.,91, 379 (1969).
J. Nagakura and M. Gouterman, J. Chem. Phys.,26, 881 (1957).
P. Chiorboli, B. Fortunato, and A. Rastelli, Ricerca Scient.,8A, 985 (1965).
Author information
Authors and Affiliations
Additional information
Translated from Izvestiya Akademii Nauk SSSR, Seriya Khimicheskaya, No. 8, pp. 1710–1714, August, 1974.
Rights and permissions
About this article
Cite this article
Épshtein, L.M., Dau, Z.F., Ashkinadze, L.D. et al. Spectral properties and structure of orthochloromercuryphenols. Russ Chem Bull 23, 1634–1637 (1974). https://doi.org/10.1007/BF00923178
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00923178