Conclusions
-
1.
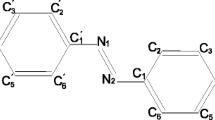
The dipole moments were determined for 18 para- and ortho-substituted phenyl benzoates in benzene and dioxane solutions. The benzene rings and phenyl benzoates have an S-trans-orientation.
-
2.
In ortho-substituted phenyl benzoates, the molecule is not coplanar. In dioxane the conformation of the molecules changes in the direction of the more coplanar structure.
Similar content being viewed by others
Literature cited
V. V. Korshak, G. L. Slonimskii, S. V. Vinogradova, A. A. Askadskii, A. V. Vasil'ev, A. V. Vasnev, and K. A. Bychko, Dokl. Akad. Nauk SSSR,199, 607 (1971).
V. V. Korshak, S. A. Pavlova, L. V. Dubrovina, V. A. Vasnev, S. V. Vinogradova, and A. V. Vasil'ev, Izv. Akad. Nauk SSSR, Ser. Khim., 1409 (1972).
B. Eda and K. Ito, Bull. Chem. Soc. Japan,29, 524 (1956);30, 164 (1957).
V. I. Minkin, O. A. Osipov, and Yu. A. Zhdanov, Dipole Moments in Organic Chemistry [in Russian], Khimiya, Leningrad (1968).
V. J. Minkin, Yu. A. Zhdanov, E. A. Medyantzeva, and Yu. A. Ostroumov, Tetrahedron,23, 3651 (1967).
Ch. H. Rao, Electronic Spectra in Chemistry [Russian translation], Mir (1964).
Author information
Authors and Affiliations
Additional information
Translated from Izvestiya Akademii Nauk SSSR, Seriya Khimicheskaya, No. 4, pp. 778–782, April, 1975.
Rights and permissions
About this article
Cite this article
Kolodyazhnyi, Y.V., Vasnev, V.A., Lapin, I.A. et al. Dipole moments and structure of derivatives of phenyl benzoate. Russ Chem Bull 24, 701–704 (1975). https://doi.org/10.1007/BF00920677
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00920677