Abstract
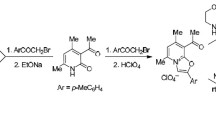
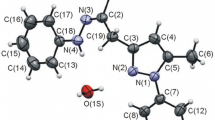
It is found that protonated pyridazine is attacked by nucleophilic acyl radicals not in the positions of lowest electron density (C-3, C-6) but in C-4 and C-5 yielding diacylpyridazines1 a-e. When acyl radicals bearing hydrogen in α-position to the carbonyl group are used, the diacylpyridazines are mainly converted to cyclopenta[d]pyridazines (e.g.2, 3, 4) by intramolecular aldol reactions. Compounds1 a-d are easily converted to 1,4-disubstituted pyridazino[4,5-d]pyridazines by condensation with semicarbazide or hydrazine respectively.
Similar content being viewed by others
Literatur
7. Mitt.:G. Heinisch undA. Mayrhofer, Mh. Chem.108, 213 (1977).
T. Caronna, G. Gardini undF. Minisci, Chem. Comm.1969, 201;
T. Caronna, G. Fronza, F. Minisci undO. Porta, J. Chem. Soc., Perkin Trans. II1972, 2035.
G. Heinisch, A. Jentzsch undM. Pailer, Mh. Chem.105, 648 (1974).
G. Heinisch und Mitarb. (unveröffentlicht)
M. Hieda, K. Omura undSh. Yurugi, Yakugaku Zasshi92, 1327 (1972); Chem. Abstr.78, 43402 k;A. Miyake, K. Itoh, N. Tada, Y. Oka undSh. Yurugi, Chem. Pharm. Bull.23, 1488 (1975);M. Tomimoto undSh. Yurugi, Takeda Kenkyusho Ho33, 151 (1974); Chem. Abstr.82, 43325;T. Matsuo undT. Miki, Yakugaku Zasshi92, 703 (1972); Chem. Abstr.77, 88417 g (1972);Y. Oka, K. Itoh, A. Miyake, N. Tada, K. Omura, M. Tomimoto undSh. Yurugi, Chem. Pharm. Bull.23, 2306, 2239 (1975);Sh. Yurugi, T. Fushimi, H. Sugihara undM. Hieda, Yakugaku Zasshi92, 1333 (1972); Chem. Abstr.78, 43400 h;K. Nishikawa, Y. Inada, H. Shimakawa, I. Kuramoto, M. Isono undS. Kikuchi, Takeda Kenkyusho32, 539 (1973); Chem. Abstr.81, 58419.
Pyridazines (R. Castle, Hrsg.), S. 4. New York: Wiley. 1973.
F. Minisci, Synthesis1973, 1.
G. Heinisch, E. Luszczak undM. Pailer, Mh. Chem.105, 763 (1974).
N. Elming, Acta Chem. Scand.6, 572 (1952).
Author information
Authors and Affiliations
Additional information
Auszugsweise vorgetragen bei den VÖCh-Chemietagen, Graz, Oktober 1977.
Rights and permissions
About this article
Cite this article
Braun, M., Hanel, G. & Heinisch, G. 4,5-Diacylpyridazine: Synthese und Umsetzung zu 1,4-Diaryl- bzw. 1,4-Dialkyl-pyridazino [4,5—d]pyridazinen 8. Mitt. über pyridazine. Monatshefte für Chemie 109, 63–71 (1978). https://doi.org/10.1007/BF00911945
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00911945