Summary
-
1.
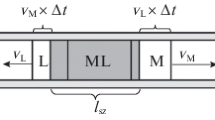
Equations have been obtained for the movement of concentration points in complexing chromatography carried out under equilibrium conditions. Basic relations connecting the rate of front movement, and the pH and concentration of the solution have been formulated.
-
2.
Expressions have been obtained which can be used in following the process of separating such closely related elements as the transuranides and rare earths. These expressions have been used to establish the conditions required for separations with various complexing agents.
-
3.
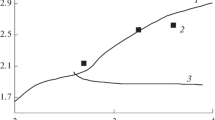
Calculations have been carried out using the data of the literature on the separation of the transuranides. It has been shown that the separation process is correctly described by the theory developed here.
Similar content being viewed by others
Literature cited
E. Tompkins, J. Khyn, and W. Cohn, J. Amer. Chem. Soc.69, 2769 (1947).
W. Cohn, G. Parker, and E. Tompkins, Nucleonics3, 22 (1948).
S. Yu. Elovich and V. N. Prussakov, Dokl. AN SSSR,112, 684 (1957).
J. Fuger, J. Inorg. and Nucl. Chem.5, 332 (1958).
S. G. Thompson, B. G. Harvey, G. R. Choppin, and G. T. Seaborg, J. Amer. Chem. Soc.76, 6229 (1954).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Elovich, S.Y. The theory of complexing chromatography and its application to the separation of closely related elements. Russ Chem Bull 11, 1454–1461 (1962). https://doi.org/10.1007/BF00907215
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00907215