Conclusions
-
1.
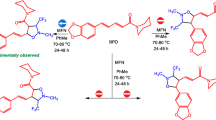
The kinetics of the intramolecular alkylation of 2-(9-iodononyl)-5-(carbethoxyacetyl)thiophene was studied in the presence of solid K2CO3.
-
2.
The intramolecular alkylationof 2-(9-iodononyl)-5-(carbethoxyacetyl) thiophene is a first-order reaction with respect to 2-(9-iodononyl)-5-(carbethoxyacetyl) thiophene, with an activation energy of 9.7 kcal per mole.
-
3.
On the basis of the principles found, it was hypothesized that the intramolecular alkylation of 2-(9-iodononyl)-5-(carbethoxyaeetyl) thiophene proceeds through a stage of formation of an anion fixed on the surface of K2CO3.
Similar content being viewed by others
Literature cited
S. Z. Taits, É. A. Krasnyanskaya, and Ya. L. Gol'dfarb, Izv. Akad. Nauk SSSR, Ser. Khim.,1968, 754.
H. Freundlich and A. Krestovnikoff, Z. Phys. Chem.,76, 79 (1911).
G. Salomon, Helv. China. Acta,19, 743 (1936).
G. Bennet, J. Chem. Soc.,1929, 2567.
G. Bennet, J. Chem. Soc.,1938, 1891.
A. Davies, M. Davies, and M. Stoll, Helv. Chim. Acta,37, 1351 (1954).
A. Brändstrom, Arkiv kemi,6, 155 (1953); Arkiv kemi,7, No. 81, 181 (1954).
H. Hunsdiecker, Ber.,75, No. 10, 1193 (1942).
S. Rhoads and A. Decora, Tetrahedron,19, 1645 (1963).
K. Cooper, E. Hughes, C. Ingold, G. Maw, and B. McNulty, J. Chem. Soc.,1948, 2051.
P. Pearson and J. Mills, J. Amer. Chem. Soc.,72, 1692 (1956).
W. Baker, J. McOmie, and W. Ollis, J. Chem. Soc.,1951, 200.
Author information
Authors and Affiliations
Additional information
Translated from Izvestiya Akademii Nauk SSSR, Seriya Khimicheskaya, No. 4, pp. 762–768, April, 1968.
Rights and permissions
About this article
Cite this article
Taits, S.Z., Krasnyanskaya, É.A. & Gol'dfarb, Y.L. A new method of synthesizing macrocyclic compounds. Russ Chem Bull 17, 735–740 (1968). https://doi.org/10.1007/BF00905742
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00905742