Abstract
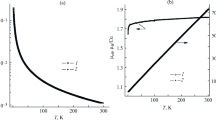
Two pairs of isomeric copper(II) chelates of the composition Cu(dm)(NO3)2 and Cu(dp)(NO3)2, resp., have been prepared (dm=6,7-dihydro-5,8-dimethyl-dibenzo[b,j][1,10]phenanthroline), anddp=6,7-dihydro-5,8-diphenyl-dibenzo[b,j][1, 10]-phenanthroline). By ESR, IR, and visible reflectance spectra the structures of the first coordination sphere of these chelates have been established, the two Cu(dm) (NO3)2 chelates and the brown α-Cu(dp) (NO3)2, having coordination number six while the green β-Cu(dp) (NO3)2 has coordination number seven. In comparison with the diamagnetically diluted complexes (solution, powder) the isomerism is found to be typical only for undiluted copper(II) complexes.
Similar content being viewed by others
Literatur
J. Gazo, Pure Appl. Chem.38, 279 (1974).
J. W. Searl, R. C. Smith undS. J. Wyard, Proc. Phys. Soc. [London]A 78, 1174 (1961).
F. K. Kneubühl, J. chem. Phys.33, 1074 (1960).
Ph. Thomas, unveröffentl. Ergebnisse.
N. F. Curtis undY. M. Curtis, Inorg. Chem.4, 804 (1965).
C. C. Addison, N. Logan, S. C. Wallwork undC. D. Garner, Quart. Rev. Chem. Soc.25, 289 (1971).
I. M. Procter, B. J. Hathaway undP. Nicholls, J. chem. Soc.A 1968, 1678.
R. J. Fereday, Bull. Chem. Soc. Japan45, 2927 (1972).
D. Rehorek undPh. Thomas, Z. Chem., im Druck.
D. L. McFadden undA. T. McPhail, J. chem. Soc. Dalton Trans.1975, 1993.
G. F. Kokoszka, C. W. Reimann undH. C. Allen, J. phys. Chem.71, 121 (1967).
D. Rehorek undPh. Thomas, Z. anorg. allg. Chem., im Druck.
E. Uhlemann, Ph. Thomas undG. Kempter, Z. anorg. allg. Chem.341, 11 (1965).
Author information
Authors and Affiliations
Additional information
Mit 2 Abbildungen
19. Mitt.:D. Rehorek, H. Spindler undPh. Thomas, Z. Chem.15, 281 (1975).
Rights and permissions
About this article
Cite this article
Rehorek, D., Ackermann, M., Spindler, H. et al. Isomere Kupfer(II)-Chelate mit Liganden vom Cuprointyp. Monatshefte für Chemie 107, 1085–1093 (1976). https://doi.org/10.1007/BF00903795
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00903795