Abstract
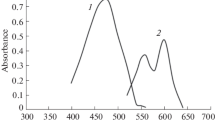
The formation of a yellow complex by the reaction between Ta(V) and 8-hydroxyquinoline-5-sulphonic acid (HQSA), has been studied spectrophotometrically in aqueous medium. At pH 6.5 the complex shows the maximum absorbance at 420 nm. TheBeer's law is obeyed by the system within the concentration range of 1 mg to 6 mg per millilitre of Ta(V).
The stoichiometry of the complex determined by the mole ratio andJob's method is 1∶2 metal-ligand. The effect of ionic strength and temperature on chelate has been studied. The interference by various ions in the system and a probable structure of the complex are also suggested.
Similar content being viewed by others
Literatur
J. L. Kassener undA. Garicarporata, Anal. Chem.27, 492 (1955).
Slive Kallaman, Treatise On Analytical Chemistry (Eds.I. M. Kolthoff undP. J. Elving), 2nd Part, Vol. 5, S. 237. New York: Interscience. 1964.
A. K. Mukherji undA. K. Dey, Anal. Chem. Acta18, 324 (1958).
A. K. Mukherji undA. K. Dey, J. Inorg. Nucl. Chem.6, 314 (1956).
J. H. Yoe undA. H. Jones, Ind. Engg. Chem. Anal. Ed.19, 11 (1944).
P. Job, Ann. Chem. Phys.8, 133 (1928).
A. E. Harvey undD. L. Manning, J. Amer. Chem. Soc.72, 4488 (1950).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Sharma, Y., Shivahare, G.C. Spektrophotometrische Bestimmung von Tantal(V) mit 8-Hydroxychinolin-5-sulfonsäure. Monatshefte für Chemie 106, 695–699 (1975). https://doi.org/10.1007/BF00902174
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00902174