Abstract
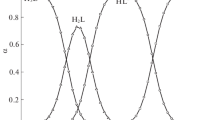
dl Aspartic acid is reduced at the d.m.e. in 0.1M tetramethylammonium bromide, tetraethylammonium bromide, sodium chloride, potassium chloride, potassium nitrate, sodium perchlorate and lithium sulphate; in aqueous media. The waves are irreversible, diffusion controlled involving one electron transfer process determined by millicoulometry. The values of the kinetic parameters, transfer coefficient (α) and formal rate constant (k° f, h) have been calculated byKoutecky's method and are 0.479 and 15.9×10−16 respectively.
Similar content being viewed by others
References
Y. Nakao andA. Nakahara, Bull. Chem. Soc. Japan46, 187 (1973), No. 1.
S. Lal, Austral. J. Chem.25, 1571 (1972), No. 7.
H. Uchiyama andS. Takamata, Nippon Kagaku Kaishi6, 1084 (1972).
T. R. Koch andW. C. Purdy, Anal. Chim. Acta54, 271 (1971).
T. De Vries andJ. L. Kroon, J. Amer. Chem. Soc.75, 2484 (1953).
J. Heyrovsky andJ. Kuta, Principles of Polarography, p. 124. New York: Academic Press. 1966.
E. E. Conn andP. K. Stumpf, Outlines of Biochemistry, p. 62. New York: J. Wiley. 1965.
J. Koutecky, Coll. Czechosl. Chem. Commun.18, 597 (1953).
L. Meites andY. Israel, J. Amer. Chem. Soc.83, 4903 (1961).
Author information
Authors and Affiliations
Additional information
With 1 Figure
Rights and permissions
About this article
Cite this article
Saxena, R.S., Saxena, M.C. Polarographic behaviour of DL aspartic acid at the D.M.E.. Monatshefte für Chemie 108, 829–833 (1977). https://doi.org/10.1007/BF00898047
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00898047