Summary
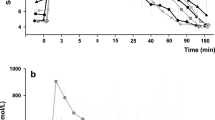
To characterize the diabetogenic effects of growth hormone, we simultaneously measured glucose turnover with 2-3H- and 6-3H-glucose in six acromegalic patients with normal fasting blood glucose and oral glucose tolerance tests. Eight healthy volunteers served as controls. All subjects were studied under both basal conditions and during glucose infusion (2 mg · kg−1 · min−1). We determined true glucose production and irreversible glucose uptake using 6-3H-glucose and glucose cycling (difference between 2-3H- and 6-3H-glucose). After an overnight fast, glucose production was higher than normal in the acromegalic patients (2.18±0.15 vs 1.85±0.03 mg · kg−1 · min−1,p < 0.05) despite hyperinsulinaemia. The metabolic clearance rate was normal. During the glucose infusion, glucose production was not suppressed as effectively in the acromegalic patients as in controls nor was glucose uptake augmented, while metabolic clearance rate was decreased. In acromegaly, basal glucose cycling was increased (0.44 ± 0.08 vs 0.25 ± 0.07 mg · kg−1 · min−1, p < 0.05). Furthermore cycling of endogenous glucose measured during glucose infusion was also augmented (0.41 ± 0.05 vs 0.24 ± 0.05 mg · kg−1 · min−1, p < 0.05). Hence the increase of glucose cycling (70%) was much more pronounced than that of glucose production (17%). In conclusion, small defects in glucose metabolism in acromegaly can be detected with sensitive tracer methods. These derangements are confined to the liver under fasting conditions, but are of both hepatic and extrahepatic origin during glucose loading.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Daughaday WH (1985) The adenohypophysis. In: Wilson JD, Foster DW (eds) Textbook of endocrinology. WB Saunders Company, Philadelphia London Toronto, p 605
Beck P, Schalch DS, Parker ML, Kipnis DM, Daughaday WH (1965) Correlative studies of growth hormone and insulin plasma concentrations with metabolic abnormalities in acromegaly. J Lab Clin Med 66: 366–379
Stinksen PH, Greenwood FC, Ellis JP, Lowy C, Rutherford A, Nabarro JDN (1967) Changes of carbohydrate tolerance in acromegaly with progress of the disease and in response to treatment. J Clin Endocrinol Metab 27: 1418–1430
Fineberg SE, Merimee TJ, Rabinowitz D, Edgar PJ (1970) Insulin secretion in acromegaly. J Clin Endocrinol Metab 30: 288–292
Trimble ER, Atkinson AB, Buchanan KD, Hadden DR (1980) Plasma glucagon and insulin concentrations in acromegaly. J Clin Endocrinol Metab 51: 626–631
Galbraith HJB, Ginsburg J, Paton A (1960) Decreased response to intra-arterial insulin in acromegaly. Diabetes 9: 459–465
Feek CM, Bevan JS, Taylor S, Brown NS, Baird JD (1981) The effect of bromocriptine on insulin secretion and glucose tolerance in patients with acromegaly. Clin Endocrinol 15: 473–478
Eastman RC, Gorden P, Roth J (1979) Conventional supervoltage irradiation is an effective treatment for acromegaly. J Clin Endocrinol Metab 48: 931–940
Muggeo M, Bar RS, Roth J, Kahn CR, Gorden P (1979) The insulin resistance of acromegaly: Evidence for two alterations in the insulin receptor on circulating monocytes. J Clin Endocrinol Met-. ab 48: 17–25
Liuzzi A, Chiodini PG, Botalla L, Cremascoli G, Siverstrini F (1972) Inhibitory effect of L-dopa on GH release in acromegaly patients. J Clin Endocrinol Metab 35: 941–943
Irie M, Tsushima T (1972) Increase of serum growth hormone concentration following thyrotropin-releasing hormone injection in patients with acromegaly or gigantism. J Clin Endocrinol Metab 35: 97–100
Efendić S, Wajngot A, Cerasi E, Luft R (1980) Insulin release, insulin sensitivity, and glucose intolerance. Proc Natl Acad Sci USA 77: 7425–7429
Hugget ASG, Nixon DA (1957) Use of glucose oxidase, peroxidase and O-dianisidine in determination of blood and urinary glucose. Lancet 2: 368–370
Herbert V, Lau KS, Gottlieb CW, Bleicher SJ (1965) Coated charcoal immunoassay of insulin. J Clin Endocrinol Metab 25: 1375–1384
Faloona GR, Unger RH (1974) Glucagon radioimmunoassay. In: Jaffe BM, Behrman HR (eds) Methods of radioimmunoassay. Academic Press, New York, pp 317–330
Dunn DF, Friedman B, Maass AR, Reichard GA, Weinhouse S (1957) Effectos of insulin on blood glucose entry and removal rates in normal dogs. J Biol Chem 225: 225–237
DeBodo RC, Steele R, Altszuler N, Dunn A, Bishop JS (1963) On the hormonal regulation of carbohydrate metabolism; studies with14C glucose. Recent Prog Horm Res 19: 445–488
Cowan HS, Hetenyi, Jr G (1971) Glucoregulatory responses in normal and diabetic dogs recorded by a new tracer method. Metabolism 20: 360–372
Radziuk J, Norwich KH, Vranić M (1978) Experimental validation of measurements of glucose turnover in nonsteady state. Am J Physiol 234 (Endocrinol Metab Gastrointest Physiol 3): E84-E93
Cherrington AD, Vranić M (1973) Effect on arginine on glucose turnover and plasma free fatty acids in normal dogs. Diabetes 22: 537–543
Bratusch-Marrain PR, Smith D, DeFronzo RA (1982) The effect of growth hormone on glucose metabolism and insulin secretion in man. J Clin Endocrinol Metab 55: 973–982
Rizza RA, Mandarino LJ, Gerich JE (1982) Effects of growth hormone on insulin action in man. Mechanisms of insulin resistance, impaired suppression of glucose production, and impaired stimulation of glucose utilization. Diabetes 31: 663–669
Bishop JS, Steele R, Altszuler N, Rathgeb I, Bjerknes C, DeBodo RC (1967) Diminished responsiveness to insulin in the growth hormone-treated normal dog. Am J Physiol 212: 272–278
Altszuler N, Rathgeb I, Winkler B, DeBodo RC, Steele R (1968) The effects of growth hormone on carbohydrate and lipid metabolism in the dog. Ann N Y Acad Sci 148: 441–458
Hue L (1981) The role of futile cycles in the regulation of carbohydrate metabolism in the liver. In: Meister A (ed) Advances in enzymology. John Wiley, New York, pp 247–331
Dunn A, Chenoweth M, Schaeffer LD (1967) Estimation of glucose turnover and the Cori cycle using glucose 6-t-14C. Biochemistry 6: 6–11
Issekutz Jr B (1977) Studies on hepatic glucose cycles in normal and methylprednisolone-treated dogs. Metabolism 26: 157–170
Katz J, Rognstad R (1976) Futile cycles in the metabolism of glucose. Curr Top Cell Regul 10: 237–289
DeFronzo RA, Gunnarsson R, Björkman O, Olsson M, Wahren J (1985) Effects of insulin on peripheral and splanchnic glucose metabolism in noninsulin-dependent (Type II) diabetes mellitus. J Clin Invest 76: 149–155
Kolterman OG, Gray RS, Griffin J, Burstein P, Insel J, Scarlett JA, Olefsky JM (1981) Receptor and postreceptor defects contribute to the insulin resistance in noninsulin-dependent diabetes mellitus. J Clin Invest 68: 957–969
Efendić S, Wajngot A, Vranić M (1985) Increased activity of the glucose cycle in the liver: Early characteristic of type 2 diabetes. Proc Natl Acad Sci 82: 2965–2969
Efendić S, Luft R, Wajngot A (1984) Aspects of the pathogenesis of type 2 diabetes. Endocrine Rev 5: 395–410
Speth M, Schulze HU (1981) Hormone-induced effects on the rat liver microsomal glucose-6-phosphatase system in vitro. Biochem Biophys Res Commun 99:134–141
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Karlander, S., Vranić, M. & Efendić, S. Increased glucose turnover and glucose cycling in acromegalic patients with normal glucose tolerance. Diabetologia 29, 778–783 (1986). https://doi.org/10.1007/BF00873216
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00873216