Abstract
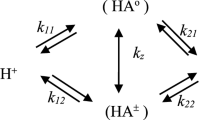
The kinetics of protonation of pseudoephedrine by 3,6-di-tert-butyl-2-hydroxyphenoxyl have been analyzed by the ESR technique. It was shown that protolysis of the radical produced closed ionic pairs with the pseudoephedrine cation in a dimer state.
Similar content being viewed by others
References
A. S. Masalimov, S. N. Nikol'skii, A. P. Abdykarimova, A. I. Prokof'ev, and A. I. Muldakhmetov,Izv. Akad. Nauk KazSSR, Ser. Khim., No. 2, 15 (1989).
A. S. Masalimov, A. I. Prokof'ev, S. P. Solodovnikov, N. N. Bubnov, and M. I. Kabachnik,Izv. Akad. Nauk SSSR, Ser. Khim., No. 4, 767 (1977).
A. Carrington and A. D. MacLachlan,Introduction to Magnetic Resonance: With Applications to Chemistry and Chemical Physics, [Russian translation], Mir, Moscow (1970), p. 266.
M. Zh. Zhurinov, A. M. Gazaliev, and S. D. Fazylov,Chemistry of Ephedrine Alkaloids [in Russian], Nauka, Alma-Ata (1990), p. 5.
A. S. Masalimov, O. D. Kemalov, É. K. Zhumadilov, and A. I. Prokof'ev,Luminescence and Study of Radiation Parameters [in Russian], Karaganda (1987), p. 74.
Additional information
A. N. Nesmeyanov Institute of Elementoorganic Compounds, Russian Academy of Sciences, 117813 Moscow. Translated from Izvestiya Akademii Nauk, Seriya Khimicheskaya, No. 9, pp. 2007–2011, September, 1992.
Rights and permissions
About this article
Cite this article
Masalimov, A.S., Nikol'skii, S.N., Abdykarimova, A.P. et al. ESR analysis of the kinetic alkalinity of pseudophedrine. Russ Chem Bull 41, 1559–1562 (1992). https://doi.org/10.1007/BF00863572
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00863572