Abstract
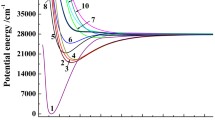
The electronic and geometric structures of the ground state and a number of excited states of the SF6 molecule and the SF −6 anion have been calculated by the discrete-variation method of the local density-functionals. The anion was found to possess a number of states stable toward the outer electron detachment, and at least one excited state stable toward dissociation. The adiabatic electron affinity (EA) was determined as 3.46 eV at the highest level of theory. This result is correlated to the high EAs of the isovalent compound SeF6 and TeF6; however, it does not agree with the presently accepted experimental estimate of 1.0 ± 0.2 eV for the SF6EA value. The basic anion configuration is octahedral with a S-F bond length of 1.717 Å. The calculated limit for the highest dissociation channel of the ground state SF −6 → SF −5 + F is ≈ 1.5 eV lower than the minimum of the total energy of the neutral molecule; this is in good agreement with experimental estimates.
Similar content being viewed by others
Literature cited
L. G. Christophorou (ed.),Electron-Molecule Interactions and Their Applications, Vols. 1, 2, Academic Press, New York (1984).
R. N. Compton, L. G. Christophorou, G. S. Hurst, and P. W. Reinhardt,J. Chem. Phys. 45, 4634 (1966).
R. K. Asundi and J. D. Craggs,Proc. Phys. Soc. 83, 611 (1964).
P. W. Harland and J. C. Thynne,J. Phys. Chem. 75, 3517 (1971).
A. V. Phelps and R. S. Van Brunt,J. Appl. Phys. 64, 4269 (1988).
A. Chutijan and S. H. Alajajin,Phys. Rev. A 31, 2885 (1985).
S. R. Hunter, J. G. Carter, and L. G. Christophorou,J. Chem. Phys. 90, 4879 (1989).
D. Spence and G. J. Schulz,J. Chem. Phys. 58, 1800 (1973).
C. L. Chen and P. J. Chantry,J. Chem. Phys. 71, 3897 (1979).
R. J. Van Brunt,J. Appl. Phys. 59, 2314 (1986).
J. K. Olthoff, R. J. Van Brunt, Y. Wang, R. L. Champion, and L. D. Doverspike,J. Chem. Phys. 91, 2261 (1989).
G. E. Streit,J. Chem. Phys. 77, 826 (1982).
E. P. Grimsrud, S. Chouwdhury, and P. Kebarle,J. Chem. Phys. 83, 1059 (1985).
P. J. Orient and A. Chutjian,Phys. Rev. A34, 1841 (1986).
E. C. M. Chen, L.-R. Shuie, E. D. D'Sa, C. F. Batten, and W. E. Wentworth,J. Chem. Phys. 88, 4711 (1988).
A. A. Christodoulides, D. L. McCorkle, and L. G. Christophorou,Electron Affinities of Atoms, Molecules, and Radicals, Vol. 2, Chap. 6 (1984).
P. J. Hay,J. Chem. Phys. 76, 502 (1982).
R. Tang and J. Callaway,J. Chem. Phys. 84, 6854 (1986).
M. Klobukowski, Z. Barandiaran, L. Seijo, and S. Huzinaga,J. Chem. Phys. 86, 1637 (1987).
E. Miyoshi, Y. Sakai, and S. Miyoshi,J. Chem. Phys. 88, 1470 (1988).
W. E. Wentworth and W. Ristau,J. Phys. Chem. 73, 2126 (1969).
J. A. D. Stockdale, R. N. Compton, and H. C. Schweinler,J. Chem. Phys. 53, 1502 (1969).
B. R. Miller and M. Fink,J. Chem. Phys. 75, 5326 (1981).
S. Lundquist and N. H. March (eds.),Theory of the Inhomogeneous Electron Gas, Plenum Press, New York (1983).
R. Fletcher,Practical Methods for Optimization Wiley, New York (1980).
L. Versluis and T. Ziegler,J. Chem. Phys. 88, 322 (1988).
E. J. Baerends, D. E. Ellis, and P. Ros,Chem. Phys. 2, 41 (1973).
J. C. Slater,Quantum Theory of Molecules and Solids, Vol. 4, McGraw-Hill, New York (1974).
C. A. J. Fletcher,Computational Galerkin Methods Springer, New York (1984).
J. G. Snijders, E. J. Baerends and P. Vernojis,At. Nucl. Data Tables 26, 486 (1982).
S. H. Vosko, L. Wilk, and M. Nusair,Can. J. Phys. 58, 1200 (1980).
A. D. Becke,Phys. Rev. A38, 3098 (1988).
A. D. Becke,J. Chem. Phys. 88, 1053 (1988).
V. Tschinke and T. Ziegler,Density Matrices and Density Functionals, R. Erdahl and V. H. Smith (eds.), Reidel, New York (1987), p. 467.
G. L. Gutsev and A. I. Bondyrev,J. Electron. Spectrosc. Relat. Phenom. 50, 103 (1990).
M. Klobukowski, S. Huzinaga, L. Seijo, and Z. Barandiaran,Theor. Chim. Acta 71, 237 (1987).
D. Hildenbrand,J. Phys. Chem. 77, 897 (1973).
D. F. McMillen and D. M. Golden,Ann. Rev. Phys. Chem. 33, 493 (1982).
T. Kiang, R. C. Estler, and R. N. Zare,J. Chem. Phys. 70, 5925 (1979).
L. M. Babcock and G. E. Streit,J. Chem. Phys. 75, 3864 (1981).
G. Herzberg,Electronic Spectra and Electronic Structure of Polyatomic Molecules Van Nostrand, Princeton (1966), p. 644.
P. Harland and J. C. J. Thynne,J. Phys. Chem. 73, 4031 (1969).
P. S. Drzaic and J. I. Brauman,J. Am. Chem. Soc. 104, 13 (1982).
S. H. Vosko and J. B. Lagowski,Density Matrices and Density Functionals, R. Erdahl and V. H. Smith (eds.), Reidel, New York (1987), p. 391.
B. I. Dunla,Ab Initio Methods in Quantum Chemistry, Vol. 2, Wiley, New York (1987), p. 287.
Y. Wang, R. L. Champion, L. D. Doverspike, J. K. Olthoff, and R. J. Van Brunt,J. Chem. Phys. 91, 2254 (1989).
C. B. Leffert, S. Y. Tang, E. W. Rothe, and T. C. Cheng,J. Chem. Phys. 61, 4929 (1974).
M. M. Hubers and J. Los,J. Chem. Phys. 63, 235 (1975).
R. N. Compton, P. W. Reinhardt, and C. D. Cooper,J. Chem. Phys. 68, 2023 (1978).
C. Lifshitz,J. Phys. Chem. 87, 3474 (1983).
M. Fenzlaff, R. Gerhard, and E. Illenberger,J. Chem. Phys. 88, 149 (1988).
L. E. Kline, D. K. Davies, C. L. Chen, and P. J. Chantry,J. Appl. Phys. 50, 6789 (1979).
J. E. Delmore and A. D. Appelhans,J. Chem. Phys. 84, 6288 (1986).
T. Ziegler and G. L. Gutsev,J. Comput. Chem. (1991) (in press).
G. L. Gutsev and A. P. Klyagina,Chem. Phys. 75, 243 (1983).
G. L. Gutsev and A. A. Levin,Chem. Phys. 51, 459 (1980).
A. K. Rappé,J. Chem. Phys. 85, 6576 (1986).
G. L. Gutsev and A. I. Boldyrev,Adv. Chem. Phys. 61, 169 (1985).
G. L. Gutsev and A. I. Boldyrev,Usp. Khim. 56, 889 (1987).
G. L. Gutsev and A. I. Boldyrev,J. Phys. Chem. 94, 2256 (1990).
Additional information
Institute for Chemical Physics, Russian Academy of Sciences, 142432 Chernogolovka. Translated from Izvestiya Akademii Nauk, Seriya Khimicheskaya, No. 3, pp. 641–649, March, 1992.
Rights and permissions
About this article
Cite this article
Gutsev, G.L. The structure of the SF6 molecule and the SF −6 anion excited states. Russ Chem Bull 41, 504–510 (1992). https://doi.org/10.1007/BF00863073
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00863073