Conclusions
-
1.
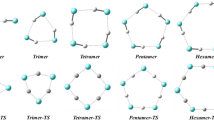
The model of associations monomer ⇌ cyclic dimer, proposed earlier for dimethylhydrazines (DMH), is incorrect.
-
2.
Association occurs according to the scheme monomer ⇌ cyclic n-mer with n=3 for 1,1-DMH and n=3 and 4 for 1,2-DMH.
-
3.
The association constants were determined for a number of temperatures, enthalpies and entropies of formation of associates, as well as average values of the hydrogen bond energy for the models considered.
Similar content being viewed by others
Literature cited
M. Saunders and J. B. Hyne, J. Chem. Phys.,29, 1319 (1958).
J. R. Crook and K. Schug, J. Amer. Chem. Soc.,86, 4271 (1964).
J. Amesley, J. Finney, and L. Sutcliff, High Resolution NMR Spectroscopy [Russian translation], Mir (1968), p. 1.
J. Biais, B. Lemanceau, and C. Lussan, J. Chim. Phys. et Phys. Chim. Biol.,64, 1019, 1030 (1967).
L. N. Erofeev and A. A. Kirnov, USSR Patent No. 265545, June 22, 1968; Byul. Izobr. No. 10, 130 (1970).
N. Muller and R. S. Reiter, J. Chem. Phys.,42, 3265 (1965).
Author information
Authors and Affiliations
Additional information
Translated from Izvestiya Akademii Nauk SSSR, Seriya Khimicheskaya, No. 6, pp. 1284–1287, June, 1972.
Rights and permissions
About this article
Cite this article
Yunda, N.G., Lagodzinskaya, G.V. & Manelis, G.B. Study of the autoassociation in hydrazines by the NMR method. Russ Chem Bull 21, 1240–1242 (1972). https://doi.org/10.1007/BF00854535
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00854535