Conclusions
-
1.
The rate constants were determined for the reaction of O-n-alkyl S-(β-ethylmercaptoethyl) methylthiophosphonates and their methyl sulfomethylates with acetylcholinesterase.
-
2.
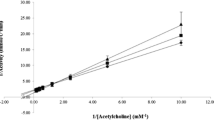
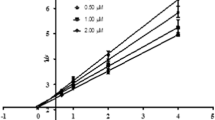
The ability of the studied compounds to react with acetylcholinesterase depends to a considerable degree on their ability to be sorbed on the active surface of the enzyme, both via hydrophobic interactions and via ionic sorption.
-
3.
The previously expressed theory that one hydrophobic segment, spatially corresponding to the n-butyl group, is present in the region of the esterase center of acetylcholinesterase was corroborated.
Similar content being viewed by others
Literature cited
A. A. Abduvakhabov, N. N. Godovikov, M. I. Kabachnik, S. S. Mikhailov, V. I. Rozengart, and V. V. Sitkevich, Izv. Akad. Nauk SSSR, Ser. Khim., 744 (1968).
A. A. Abduvakhabov, V. L. Avdeeva, N. N. Godovikov, M. I. Kabachnik, A. B. Katsnel'son, S. S. Mikhailov, V. I. Rozengart, R. V. Sitkevich, and Ya. S. Smusin, Izv. Akad. Nauk SSSR, Ser. Khim., 2033 (1968).
A. A. Abduvakhabov, N. N. Godovikov, M. I. Kabachnik, V. I. Rozengart, and R. V. Sitkevich, Izv. Akad. Nauk SSSR, Ser. Khim., 1117 (1969).
A. P. Brestkin and D. L. Pevzner, Biokhimiya,31, 1174 (1966).
W. Berry, Biochem. J.,49, 615 (1951).
I. L. Brik, Biokhimiya,34, 90 (1969).
A. A. Abduvakhabov, N. N. Godovikov, M. I. Kabachnik, S. S. Mikhailov, V. I. Rozengart, and R. V. Sitkevich, Dokl. Akad. Nauk SSSR,171, 857 (1966).
M. I. Kabachnik and N. N. Godovikov, Dokl. Akad. Nauk SSSR,196, 348 (1971).
A. P. Brestkin, R. I. Volkova, N. N. Godovikov, and M. I. Kabachnik, Izv. Akad. Nauk SSSR, Ser. Khim. 2028 (1968).
Author information
Authors and Affiliations
Additional information
Translated from Izvestiya Akademii Nauk SSSR, Seriya Khimicheskaya, No. 5, pp. 1169–1172, May, 1972.
Rights and permissions
About this article
Cite this article
Abduvakhabov, A.A., Godovikov, N.N., Kabachnik, M.I. et al. Reaction of O-n-alkyl S-(β-ethylmercaptoethyl) methylthiophosphonates and their methyl sulfomethylates with acetylcholinesterase. Russ Chem Bull 21, 1117–1120 (1972). https://doi.org/10.1007/BF00853782
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00853782