Abstract
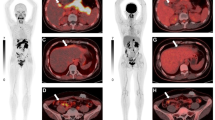
Iodine-131 3F8, a murine IgG3 monoclonal antibody that targets to GD2-bearing tumors, was administered intravenously to 12 patients with brain tumors. Six patients received 2 mCi (0.74 Bq) of131I-3F8, five patients 10 mCi (3.7 Bq)/1.73 m2 of131I-3F8, and one patient 2.6 mCi (0.96 Bq) of124I-3F8, with no side-effects. Nine of 11 malignant gliomas and the single metastatic melanoma showed antibody localization, with the best tumor delineation on single-photon emission tomography (SPET) following 10 mCi (3.7 Bq)/1.73 m2 dose. No nonspecific uptake in the normal craniospinal axis was detected. There was no difference in the pharmacokinetics of low-dose versus the higher-dose antibody groups; plasma and total-body half-lives were 18 h and 49 h, respectively. Surgical sampling and time-activity curves based on quantitative imaging showed peak uptake in high-grade glioma at 39 h, with a half-life of 62 h. Tumor uptake at time of surgery averaged 3.5×10−3 %ID/g and peak activity by the conjugate view method averaged 9.2×10−3 %ID/g (3.5–17.8). Mean radiation absorption dose was 3.9 rad per mCi injected (range 0.7–9.6) or 10.5 cGy/Bq (range 1.9–26). There was agreement on positive sites when immunoscintigraphy was compared with technetium-99m glucoheptonate/diethylene triamine penta-acetic acid planar imaging, thallium-201 SPET, and fluorine-18 fluorodeoxyglucose positron emission tomography. Taken together, these data suggest that quantitative estimates of antibody targeting to intracranial tumors can be made using the modified conjugate view method.
Similar content being viewed by others
References
Walker MD, Green S, Byar D, et al. Randomized comparisons of radiotherapy and nitrosoureas for the treatment of malignant glioma after surgery.N Engl J Med 1980;303:1323–1329.
Zalutsky MR, Moseley RP, Benjamin JC, et al. Monoclonal antibody and F(ab')2 fragment delivery to tumor in patients with glioma: comparison of intracarotid and intravenous administration.Cancer Res 1990;50:4105–4110.
Lashford LS, Davies AG, Richardson RB, et al. A pilot study of 1 311 monoclonal antibodies in the therapy of leptomeningeal tumors.Cancer 1988;61:857–868.
Riethmiiller G, Schneider-Gädicke E, Schlimok G, et al. Randomised trial of monoclonal antibody for adjuvant therapy of resected Dukes' C colorectal carcinoma.Lancet 1994;343:1177–1183.
Kemshead JT, Papanastassiou V, Coakham HB, Pizer BL. Monoclonal antibodies in the treatment of central nervous system malignancies.Eur J Cancer 1992;28:511–513.
Press OW, Eary JF, Appelbaum FR, et al. Radiolabeled antibody therapy of B-cell lymphoma with autologous bone marrow support. N Engl J Med 1993;329:1219–1224.
Schwartz MA, Lovett DR, Redner A, et al. Dose-escalation trial of M195 labeled with iodine 131 for cytoreduction and marrow ablation in relapsed or refractory myeloid leukemias.J Clin Oncol 1993;11:294–303.
Kaminski MS, Zasadny KR, Francis IR, et al. Radioimmunotherapy of B-cell lymphoma with 131-I-anti-B l-(anti-CD20) antibody.N Engl J Med 1993;329:459–465.
Urch CE, George AJT, Stevenson GT, et al. Intra-thecal treatment of leptomeningeal lymphoma with immunotoxin.Int J Cancer 1991;47:909–915.
Rodden FA, Wiegandt H, Bauer BL. Gangliosides: the relevance of current research to neurosurgery.J Neurosurg 1991; 74:606–619.
Valentino LA, Ladisch S. Localization of shed human tumor gangliosides: association with serum lipoproteins.Cancer Res 1992;52:810–814.
Berra B, Gaini SM, Riboni L. Correlation between ganglioside distribution and histological grading of human astrocytoma.Int J Cancer 1985;36:363–366.
Larson SM, Sgouros G, Cheung NK. Antibodies in cancer treatment: radioisotope conjugates. In: DeVita VT, Helluran S, Rosenberg SA. eds.Biologic therapy of cancer: principles and practice, 2nd edn. Philadelphia: J.B. Lippincott; 1995:534–552.
Wikstrand CJ, Fredman P, Svennerholm L, et al. Expression of gangliosides GM2, GD2, GD3, 3′-sioLM1, and 3′,6′ isoLD1 in CNS malignancies as defined by epitope-characterized monoclonal antibodies (Mabs) [abstract]. 9th International Conference on Brain Tumors Research and Therapy 1991.
Lougee DC, Wikstrand CJ, Mansson JE, et al. Disialoganglioside GD2 in human neuroectodermal tumor cell lines and gliomas.Acta Neuropathol (Berl) 1991;82:45–54.
Hoon DS, Banez M, Okun E, Morton DL, Irie RE Modulation of human melanoma cells by interleukin-4 and in combination with gamma-interferon or alpha-tumor necrosis factor.Cancer Res 1991;51:2002–2008.
Saito M, Yu RK, Cheung NKV. Ganglioside GD2 specificity of monoclonal antibodies to human neuroblastoma cell.Biochem Biophys Res Commun 1985;127:1–4.
Ye JN, Cheung NKV. A novel O-acetylated ganglioside detected by anti-GD2 monoclonal antibodies.Int J Cancer 1992;50:197–201.
Cheung NK, Landmeier B, Neely J, et al. Complete tumor ablation with iodine 131-radiolabeled disialoganglioside GD2 specific monoclonal antibody against human neuroblastoma xenografted in nude mice.J Natl Cancer Inst 1986;77: 739–745.
Yeh SD, Larson SM, Burch L, et al. Radioimmunodetection of neuroblastoma with iodine-131-3F8: correlation with biopsy, iodine-131-metaiodobenzylguanidine (MIBG) and standard diagnostic modalities.J Nucl Med 1991;32:769–776.
Wu Z, Schwartz E, Seeger RC, Ladisch S. Expression of GD2 ganglioside by untreated primary human neuroblastomas.Cancer Res 1986;46:440–443.
Cheung NK, Lazarus H, Miraldi FD, et al. Ganglioside GD2 specific monoclonal antibody 3F8 — a phase I study in patients with neuroblastoma and malignant melanoma.J Clin Oncol 1987;5:1430–1440.
Miraldi FD, Nelson AD, Kraly C, et al. Diagnostic imaging of human neuroblastoma with radiolabeled antibody.Radiology 1986;161:413–418.
Lindmo T, Boven E, Cuttitta F, Fedorko J, Bunn PA. Determination of the immunoreactive fraction of radiolabeled monoclonal antibodies by linear extrapolation to binding at infinite antigen excess.J Immunol Methods 1984;72:77–89.
Daghighian F, Pentlow KS, Larson SM, et al. Development of a method to measure kinetics of radiolabeled monoclonal antibody in human tumors with applications to microdosimetry: positron emission tomography studies of iodine-124 labeled 3F8 monoclonal antibody in glioma.Eur J Nucl Med 1993; 20:402–409.
Thomas SR, Maxon HP, Kereiakes JG. In vivo quantitation of lesion radioactivity using external counting methods.Med Phys 1976;3:253–255.
Nelson AD, Miraldi F, Cheung NKV. Biodistribution and dosimetry of 3F8 neuroblastoma monoclonal antibody.Am J Physiol Imaging 1989;4:143–150.
Lammie GA, Cheung NKV, Gerald W, Rosenblum M, Cordon-Cardo C. Ganglioside GD2 expression in the human nervous system and in neuroblastomas — an immunohistochemical study.Int J Oncol 1993;3:909–915.
Jain RK, Baxter LT. Mechanisms of heterogeneous distribution of monoclonal antibodies and other macromolecules in tumors: significance of elevated interstitial pressure.Cancer Res 1988;48:7022–7032.
Taguchi A. Clinical significance of thallium-210 single photon emission computerized tomography (T1-201 SPET) in the evaluation of viability of gliomas.Kurume Med J 1992;39: 267–278.
Oriuchi N, Tamura M, Shibazaki T, et al. Evaluation of 201T1 SPET in patients with glioma: a comparative study with histological diagnosis, clinical feature and proliferative activity.Kaku Igaku 1991;28:1263–1271.
Larson SM, Pentlow KS, Volkow ND, et al. PET scanning of iodine-124-3F8 as an approach to tumor dosimetry during treatment planning for radioimmunotherapy in a child with neuroblastoma.J Nucl Med 1992;33:2020–2023.
Epenetos AA, Snook D, Durbin H, Johnson PM, Taylor-Papadimitriou J. Limitations of radiolabeled monoclonal antibodies for localization of human neoplasms.Cancer Res 1986;46:3183–3191.
Moseley R, Zalutsky MR, Coakham HB, Coleman RE, Bigner DD. Distribution of131I 81C6 monoclonal antibody (Mab) administered via carotid artery in patients with glioma (G). J Nucl Med 1987;28:603.
Zalutsky MR, Moseley RP, Coakham HB, Coleman RE, Bigner DD. Pharmacokinetics and tumor localization of131I-labeled anti-tenascin monoclonal antibody 81c6 in patients with gliomas and other intracranial malignancies.Cancer Res 1989;49:2807–2813.
Richardson RB, Davies AG, Bourne SP, et al. Radioimmunolocalization of human brain tumors. Biodistribution of radiolabelled monoclonal antibody UJ13A.Eur J Nucl Med 1986; 12:313–320.
Colcher D, Bird R, Roselli M, et al. In vivo tumor targeting of a recombinant single-chain antigen-binding protein.J Natl Cancer Inst 1990;82:1191–1197.
Riva P, Arista A, Seccamani E, et al. Improved tumor targeting by direct intralesional injection of radiolabeled monoclonal antibody: a phase I study in brain glioma.J Nucl Med 1991;32:922.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Arbit, E., Cheung, NK.V., Yeh, S.D.J. et al. Quantitative studies of monoclonal antibody targeting to disialoganglioside GD2 in human brain tumors. Eur J Nucl Med 22, 419–426 (1995). https://doi.org/10.1007/BF00839056
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00839056