Abstract
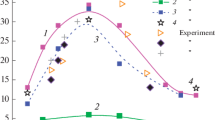
Based on a review of four mechanisms for the chemical kinetics in the study of controlled combustion of CS2+O2 mixtures, expressions were derived, based on the stoichiometric composition, relating the velocity of propagation of a laminar flame and the temperature and pressure of the mixture. The expressions were in good agreement with the experimental data.
Similar content being viewed by others
Literature Cited
V. N. Kondrat'ev, Kinetics and Catalysis,13, No. 6, 1367 (1972).
V. V. Azatyan, B. V. Novozhilov, V. S. Posvyanskii, et al., Kinetics and Catalysis,17, No. 6, 1386 (1976).
K. N. Homann, G. Krome, and H. G. ragner, Berichte Bunsenges Phys. Chem.,74, 654 (1970).
V. G. Voronkov and N. N. Semenov, Zh. Fiz. Khim.,13, 1695 (1939).
V. A. Dudkin, and V. B. Librovich, Chem. Phys.,5, No. 4, 528 (1986).
A. A. Vetter and F. E. C. Culick, Combust. Flame,30, 107 (1977).
Additional information
Moscow. Translated from Fizika Goreniya i Vzryva, Vol. 27, No. 6, pp. 60–62, November–December, 1991.
Rights and permissions
About this article
Cite this article
Dudkin, V.A. Relation between velocity of propagation of isothermal flame and parameters of CS2+O2 mixtures. Combust Explos Shock Waves 27, 704–706 (1991). https://doi.org/10.1007/BF00814513
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00814513