Abstract
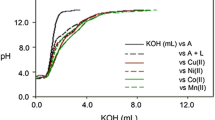
The mixed-ligand complexes of thorium(IV) with quinizarin (quin) and as a second ligand,L, salicyclic acid (sa), thiosalicylic acid (tsa) or anthranilic acid (ant) were studied potentiometrically in 40% (v/v) ethanol-water medium [I=100 mmol dm−3 (KNO3), 25±0.1 °C]. The equilibria existing in solutions were demonstrated and the ternary stability constants of the 1:1:1 Th-quin-L-complexes were characterized. All of these biligand complexes are considerably more stable than the corresponding monoligand ones. In addition, the relatively most stable ternary complex is formed withant which is the best π-acceptor. The order of stability of the ternary complexes is in accordance with the calculated π-charge densities of the varying ligating group in the ligandL. The values of the equilibrium constants (log units) for the reaction: Th(quin)2+ThL 2⇌2Th(quin)(L) are 2.47 (0.13), 2.60 (0.3) and 4.25 (0.86) for Th(quin)(tsa), Th(quin)(sa) and Th(quin)(ant), respectively. The constants given in parentheses correspond to Δ logK Th (= logK )(L)Th(quin)Th(quin — logK ThThL ).
Zusammenfassung
Die gemischtligandigen Komplexe von Thorium(IV) mit Chinizarin (quin) und als zweiten Liganden (L) Salizylsäure (sa), Thisalizylsäure (tsa) oder Anthranilsäure (ant) wurden potentiometrisch in 40% (v/v) Ethanol-Wasser [I=100 mmol dm−3 (KNO3),t=25±0.1 °C] untersucht. Die vorliegenden Gleichgewichte wurden formuliert und die ternären Stabilitätskonstanten der 1:1:1 Th-quin-L-Komplexe bestimmt. Alle zweiligandigen Komplexe sind wesentlich stabiler als die entsprechenden Monoligand-Komplexe. Der relativ stabilste Komplex wurde mitant gebildet, daant der beste π-Acceptor ist. Die Reihung der relativen Stabilitäten stimmt mit den berechneten π-Elektronendichten der verschiedenen LigandenL überein. Die Werte der Gleichgewichtskonstanten (in log Einheiten) für die Reaktion Th(quin)2+ThL 2⇌2Th(quin)(L) sind 2.47 (0.13), 2.60 (0.30) and 4.25 (0.86) für Th(quin)(tsa), Th(quin)(sa) bzw. Th(quin)(ant), wobei die Werte in Klammern ΔlogK Th = logK )(L)/Th(quin)Th(quin — logK ThThL entsprechen.
Similar content being viewed by others
References
Luo QH, Meng JX (1980) Kao Teng Hsueh Hsiao Hua Hsueh Pao, 1: 17
Ramalingam K, Krishnamoorthy CR (1982) Inorg Chim Acta 67: 167
Sharma CL, Jain PK (1977) Ind J Chem 15(A): 110
Sitaramalakshmi C, Venkateswarlu C (1975) Ind J Chem 13: 625
Pachauri OP, Tandon JP (1976) Monatsh Chem 107: 991
Pachauri OP, Tandon JP (1975) Z Natur Anorg Chem 30 B: 751;Pachauri OP, Tandon JP (1975) J Inorg Nucl Chem 27: 2321;Pachauri OP, Tandon JP (1976) Monatsh Chem 107: 83;Pachauri OP, Tandon JP (1977) Ind J Chem 15: 1
Nayan R (1981) J Inorg Nucl Chem 43: 3238
Idriss KA, Seleim MM, Abu-Baker MS, Saleh MS (1982) Analyst 107: 12
Idriss KA, Seleim MM, Abu-Baker MS, Saleh MS (1982) Ind J Chem 21 A: 395
Idriss KA, Seleim MM, Abu-Baker MS, Saleh MS (1984) Annali Chimia 74: 845
Idriss KA, Seleim MM, Hassan MK, Abu-Baker MS, Sedaira H (1985) Analyst 110: 705
Seleim MM, Idriss KA, Saleh MS, Hashem EY (1986) Analyst 111: 677
Seleim MM, Idriss KA, Saleh MS, Hashem EY (1986) Polyhedron 5: 1525
Irving HM, Rossotti HS (1953) J Chem Soc 1953: 3397; 1954: 2904
Chidambaram MV, Bhattacharya PK (1970) J Inorg Nucl Chem 32: 3271
Agrawal BH, Dwivedi K, Chandra M, Agrawala B, Dey KA (1977) J Ind Chem Soc 54: 931
Pariser R, Parr RC (1953) J Chem Phys 21: 767;Pople JA (1953) Trans Faraday Soc 49: 1375;Pariser R (1956) J Chem Phys 24: 250
Nishimoto K, Forster LS (1968) Bull Chem Soc Jpn 41: 2254
Idriss KA, Hassan MK, Abu-Baker MS, Sedaira H (1984) Analyst 109: 1389
Issa IM, Issa RM, Idriss KA, Hammam AM (1976) Ind J Chem 14(B): 117
Sigel H (1975) Angew Chem 87: 391; (1975) Angew Chem Int Ed Engl 14: 394
Dewitt R, Watters JI (1954) J Am Chem Soc 76: 3810
Kida S (1956) Bull Chem Soc Jpn 29: 805;Ramakrishna RS, Fernandopulla ME (1970) J Inorg Nucl Chem 33: 1940
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Idriss, K.A., Seleim, M.M., El-Shahawy, A.S. et al. Effect of the varying π-accepting properties of someortho-substituted benzoic acids on the stability of mixed-ligand complexes also containing quinizarin and thorium(IV). Monatsh Chem 119, 683–691 (1988). https://doi.org/10.1007/BF00809681
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00809681