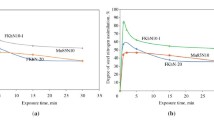
Conclusions
-
1.
A study was made of the kinetics of the reaction between chromium carbide and molten copper-nickel alloys containing 20 and 40% nickel.
-
2.
The dissolution rate of chromium carbide in the molten alloys investigated is controlled by diffusional transport in the liquid phase.
-
3.
The dissolution rate of chromium carbide in a molten Cu — 20% Ni alloy is determined by the diffusional characteristics of carbon in the alloy. The diffusion coefficient of carbon in molten copper-nickel alloys containing 20% Ni was calculated.
-
4.
During the dissolution of the higher chromium carbide in a molten Cu — 20% Ni alloy, a graphite layer forms at the interface of the two phases. No graphite deposition is observed during the dissolution of the carbide in a molten Cu — 40% Ni alloy.
Similar content being viewed by others
Literature cited
G. A. Yasinskaya, Poroshkovaya Met., No. 7, 53 (1966).
G. V. Samsonov, A. D. Panasyuk, and G. K. Kozina, Poroshkovaya Met., No. 11, 42 (1968).
V. N. Eremenko, L. N. Mekhanoshina, and M. M. Churakov, Fiz.-Khim. Mekhan. Mat., No. 2 (1971).
V. N. Eremenko and M. M. Churakov, Fiz.-Khim., Mekhan. Mat., No. 3 (1970).
V. G. Levich, Physicochemical Hydrodynamics [in Russian], Fizmatgiz (1959).
A. A. Vertman and A. M. Samarin, Dokl. Akad. Nauk SSSR,132, No. 3, 572 (1960).
V. N. Eremenko and M. M. Churakov, Fiz.-Khim. Mekhan. Mat., No. 5 (1971).
I. A. Cahill and A. D. Kirshenbaum, J. Phys. Chem.,66, No. 2, 1080 (1962).
E. S. Filippov and A. M. Samarin, Physicochemical Principles of Steelmaking [in Russian], Nauka (1968).
Author information
Authors and Affiliations
Additional information
Translated from Poroshkovaya Metallurgiya, No. 9 (117), pp. 48–51, September, 1972.
Rights and permissions
About this article
Cite this article
Eremenko, V.N., Churakov, M.M. Reaction of chromium carbide with molten copper-nickel alloys. Powder Metall Met Ceram 11, 724–726 (1972). https://doi.org/10.1007/BF00801270
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00801270