Abstract
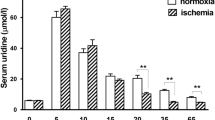
Hearts preconditioned by brief ischemia are characterized by a reduced rate of cellular purine metabolite production during subsequent prolonged ischemia; the purpose of this study was to determine if transient exogenous adenosine pretreatment can mimic this phenomenon. The accumulation of interstitial fluid (ISF) purine metabolites during prolonged ischemia in untreated anesthetized dogs (n=7) was compared to that in a group pretreated with brief ischemia (ischemic preconditioned group; n=9), a group pretreated with 1.5 μmoles/min intracoronary adenosine (n=7). Ischemic preconditioning was achieved by a 5 min period of left anterior descending coronary artery (LAD) occulusion followed by 10 min of reperfusion. The adenosine-treated groups were subjected to 10 min of intracoronary adenosine followed by 10 min of recovery. All animals were exposed to 60 min LAD occlusion followed by 60 min reperfusion. The changes in ISF adenosine and adenosine metabolites were assessed by cardiac microdialysis, using dialysate concentrations as indices of ISF levels. Ischemic preconditioning decreased the rate of dialysate adenosine and total purine accumulation during the prolonged ischemia. Although the two doses of exogenous adenosine bracketed the increase in ISF adenosine seen with ischemic preconditioning, neither adenosine dose was able to attenuate the rate of purine metabolite accumulation during prolonged ischemia. We conclude that exogenous adenosine pretreatment is unable to mimic the reduced ischemia-induced purine efflux that is characteristic of myocytes pretreated with brief ischemia.
Similar content being viewed by others
References
Auchampach JA, Gross GJ (1993) Adenosine A1 receptors, KATP channels, and ischemic preconditioning in dogs. Am J Physiol 264: H1327-H1336
Cave AC, Hearse DJ (1992) Ischaemic preconditioning and contractile function: studies with normothermic and hypothermic global ischaemia. J Mol Cell Cardiol 24: 1113–1123
Delyani JA, Van Wylen DGL (1994) Endocardial and epicardial intersititial purines and lactate during graded ischemia. Am J Physiol 266: H1019-H1026
Finegan BA, Lopaschuk GD, Gandhi M, Clanachan AS (1995) Ischemic preconditioning inhibits glycolysis and proton production in isolated working rat hearts. Am J Physiol 269: H1767-H1775
Goto M, Cohen MV, Van Wylen DGL, Downey JM (1996) Attenuated purine production during subsequent ischemia in preconditioned rabbit myocardium is unrelated to the mechanism of protection. J Mol Cell Cardiol 28: 447–454
Hagar JM, Hale SL, Kloner RA (1991) Effect of preconditioning ischemia on reperfusion arrhythmias after coronary artery occlusion and reperfusion in the rat. Circ Res 68: 61–68
Headrick JP (1996) Ischemic Preconditioning: bioenergetic and metabolic changes and the role of endogenous adenosine. J Mol Cell Cardiol 28: 1227–1240
Kida M, Fujiwara H, Ishida M, Kawai C, Ohura M, Miura I, Yabuuchi Y (1991) Ischemic preconditioning preserves creatine phosphate and intracellular pH. Circulation 84: 2495–2503
Lasley RD, Konyn PJ, Hegge JO, Mentzer RM (1995) Effects of ischemia and adenosine preconditioning on interstitial fluid adenosine and myocardial infarct size. Am J Physiol 269: H1460-H1466
Lawson CS, Coltart DJ, Hearse DJ (1993) “Dose”-dependency and temporal characteristics of protection by ischaemic preconditioning against ischaemia-induced arrhythmias in rat hearts. J Mol Cell Cardiol 25: 1391–1402
Liu GS, Richards SC, Olsson RA, Mullane K, Walsh RS, Downey JM (1994) Evidence that the Adenosine A3 receptor may mediate the protection afforded by preconditioning in the isolated rabbit heart. Cardiovasc Res 28: 1057–61
Liu GS, Thornton J, Van Winkle DM, Stanley AWH, Olsson RA, Downey JM (1991) Protection against infarction afforded by preconditioning is mediated by A1 adenosine receptors in rabbit heart. Circulation 84: 350–56
Murry CE, Jennings RB, Reimer KA (1986) Preconditioning with ischemia: a delay of lethal cell injury in ischemic myocardium. Circulation 74: 1124–36
Murry CE, Richard VJ, Reimer KA, Jennings RB (1990) Ischemic preconditioning slows energy metabolism and delays ultrastructural damage during a sustained ischemic episode. Circ Res 66: 913–931
Schaefer S, Carr LJ, Prussel E, Ramasamy R (1995) Effects of glycogen depletion on ischemic injury in isolated rat hearts: insights into preconditioning. Am J Physiol 268: H935-H944
Shiki K, Hearse DJ (1987) Preconditioning of ischemic myocardium: reperfusion-induced arrhythmias. Am J Physiol 253: H1470-H1476
Silva PH, Dillon D, Van Wylen DGL (1995) Adenosine deaminase inhibition augments interstitial adenosine but does not attenuate myocardial infarction. Cardiovasc Res 29: 616–623
Tsuchida A, Liu Y, Liu GS, Cohen MV, Downey JM (1994) 377-1 agonists precondition rabbit ischemic myocardium independent of adenosine by direct activation of protein kinase C. Circ Res 75:576–585
Van Wylen DGL (1994) Effect of ishemic preconditioning on interstitial purime metabolite and lactate accumulation during myocardial ischemia. Circulation 89: 2283–89
Van Wylen DGL, Willis J, Sodhi J, Weiss RJ, Lasley RD, Mentzer RM (1990) Cardiac microdialysis to estimate interstitial adenosine and coronary blood flow. Am J Physiol 258: H1642-H1649
Yao Z, Gross GJ (1994) A comparison of adenosine-induced cardioprotection and ischemic preconditioning in dogs: efficacy, time course, and role of KATP channels. Circulation 89: 1229–36
Young W (1980) H2 clearance measurements of blood flow: review of technique and polarographic principles. Stroke 11: 552–564
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Van Wylen, D.G.L., Manthei, S.A. Purine metabolite accumulation during myocardial ischemia: Adenosine pretreatment versus brief ischemia. Basic Res Cardiol 92, 368–377 (1997). https://doi.org/10.1007/BF00796210
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00796210