Abstract
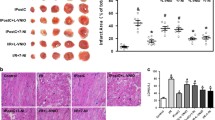
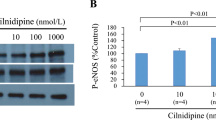
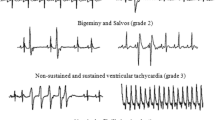
We and others have demonstrated a protective role for pacing postconditioning (PPC) against ischemia/reperfusion (I/R) injury in the heart; however, the underlying mechanisms behind these protective effects are not completely understood. In this study, we wanted to further characterize PPC-mediated cardiac protection, specifically identify optimal pacing sites; examine the role of oxidative stress; and test the existence of a potential synergistic effect between PPC and adenosine. Isolated rat hearts were subjected to coronary occlusion followed by reperfusion. PPC involved three, 30 s, episodes of alternating left ventricular (LV) and right atrial (RA) pacing. Multiple pacing protocols with different pacing electrode locations were used. To test the involvement of oxidative stress, target-specific agonists or antagonists were infused at the beginning of reperfusion. Hemodynamic data were digitally recorded, and cardiac enzymes, oxidant, and antioxidant status were chemically measured. Pacing at the LV or RV but not at the heart apex or base significantly (P < 0.001) protected against ischemia-reperfusion injury. PPC-mediated protection was completely abrogated in the presence of reactive oxygen species (ROS) scavenger, ebselen; peroxynitrite (ONOO−) scavenger, uric acid; and nitric oxide synthase inhibitor, L-NAME. Nitric oxide (NO) donor, snap, however significantly (P < 0.05) protected the heart against I/R injury in the absence of PPC. The protective effects of PPC were significantly improved by adenosine. PPC-stimulated protection can be achieved by alternating LV and RA pacing applied at the beginning of reperfusion. NO, ROS, and the product of their interaction ONOO− play a significant role in PPC-induced cardiac protection. Finally, the protective effects of PPC can be synergized with adenosine.






Similar content being viewed by others
References
Arstall MA, Sawyer DB, Fukazawa R, Kelly RA (1991) Cytokine-mediated apoptosis in cardiac myocytes: the role of inducible nitric oxide synthase induction and peroxynitrite generation. Circ Res 85:829–840
Babiker FA, Hoteit LJ, Joseph S, Mustafa AS, Juggi JS (2012) The role of 17-beta estradiol in ischemic preconditioning protection of the heart. Exp Clin Cardiol 17:95–100
Babiker FA, Joseph S, Juggi J (2014) The protective effects of 17beta-estradiol against ischemia-reperfusion injury and its effect on pacing postconditioning protection to the heart. J Physiol Biochem 70:151–162
Babiker FA, Lorenzen-Schmidt I, Mokelke E, Vanagt WY, Delhaas T (2010) Waltenberger J, Cleutjens JP, and Prinzen FW. Long-term protection and mechanism of pacing-induced postconditioning in the heart. Basic Res Cardiol 105:523–533
Baines CP, Kaiser RA, Sheiko T, Craigen WJ, Molkentin JD (2007) Voltage-dependent anion channels are dispensable for mitochondrial-dependent cell death. Nat Cell Biol 9:550–555
Beauloye C, Bertrand L, Horman S, Hue L (2011) AMPK activation, a preventive therapeutic target in the transition from cardiac injury to heart failure. Cardiovasc Res 90:224–233
Blanc JJ, Etienne Y, Gilard M et al. KG 1997 Evaluation of different ventricular pacing sites in patients with severe heart failure: results of an acute hemodynamic study. Circulation 96: 3273–3277.
Brown GC, Borutaite V (2007) Nitric oxide and mitochondrial respiration in the heart. Cardiovasc Res 75:283–290
Chen YR, Zweier JL (2014) Cardiac mitochondria and reactive oxygen species generation. Circ Res 114:524–537
Cohen MV, Downey JM (2011) Ischemic postconditioning: from receptor to end-effector. Antioxid Redox Signal 14:821–831
Costa AD, Jakob R, Costa CL, Andrukhiv K, West IC, Garlid KD (2006) The mechanism by which the mitochondrial ATP-sensitive K+ channel opening and H2O2 inhibit the mitochondrial permeability transition. J Biol Chem 281:20801–20808
Erel O (2005) A new automated colorimetric method for measuring total oxidant status. Clin Biochem 38:1103–1111
Erel O (2004) A novel automated direct measurement method for total antioxidant capacity using a new generation, more stable ABTS radical cation. Clin Biochem 37:277–285
Fan Y, Yang S, Zhang X, Cao Y, Huang Y (2012) Comparison of cardioprotective efficacy resulting from a combination of atorvastatin and ischaemic post-conditioning in diabetic and non-diabetic rats. Clin Exp Pharmacol Physiol 39:938–943
Ferrera R, Benhabbouche S, Bopassa JC, Li B, Ovize M (2009) One hour reperfusion is enough to assess function and infarct size with TTC staining in Langendorff rat model. Cardiovasc Drugs Ther 23:327–331
Folino A, Losano G, Rastaldo R (2013) Balance of nitric oxide and reactive oxygen species in myocardial reperfusion injury and protection. J Cardiovasc Pharmacol 62:567–575
Freixa X, Bellera N, Ortiz-Perez JT et al (2012) Ischaemic postconditioning revisited: lack of effects on infarct size following primary percutaneous coronary intervention. Eur Heart J 33:103–112
Hahn JY, Song YB, Kim EK et al (2013) Ischemic postconditioning during primary percutaneous coronary intervention: the effects of postconditioning on myocardial reperfusion in patients with ST-segment elevation myocardial infarction (POST) randomized trial. Circulation 128:1889–1896
Hausenloy DJ, Yellon DM (2009) Preconditioning and postconditioning: underlying mechanisms and clinical application. Atherosclerosis 204:334–341
Heusch G (2004) Postconditioning: old wine in a new bottle? J Am Coll Cardiol 44:1111–1112
Jeon YT, Hwang JW, Lim YJ, Kim AN, Park HP (2013) A combination of sevoflurane postconditioning and albumin increases Bcl-2 expression after transient global cerebral ischemia compared with either sevoflurane postconditioning or albumin alone. J Neurosurg Anesthesiol 25:43–50
Kupai K, Csonka C, Fekete V et al (2009) Cholesterol diet-induced hyperlipidemia impairs the cardioprotective effect of postconditioning: role of peroxynitrite. Am J Physiol Heart Circ Physiol 297:H1729–H1735
Layland J, Carrick D, Lee M, Oldroyd K, Berry C (2014) Adenosine: physiology, pharmacology, and clinical applications. JACC Cardiovasc Interv 7:581–591
Lebuffe G, Schumacker PT, Shao ZH, Anderson T, Iwase H, Vanden Hoek TL (2003) ROS and NO trigger early preconditioning: relationship to mitochondrial KATP channel. Am J Physiol Heart Circ Physiol 284:H299–H308
Li J, Loukili N, Rosenblatt-Velin N, Pacher P, Feihl F, Waeber B, Liaudet L (2013) Peroxynitrite is a key mediator of the cardioprotection afforded by ischemic postconditioning in vivo. PLoS One 8:e70331
Limalanathan S, Andersen GO, Klow NE, Abdelnoor M, Hoffmann P, Eritsland J (2014) Effect of ischemic postconditioning on infarct size in patients with ST-elevation myocardial infarction treated by primary PCI results of the POSTEMI (POstconditioning in ST-elevation myocardial infarction) randomized trial. J Am Heart Assoc 3:e000679
Lonborg JT (2015) Targeting reperfusion injury in the era of primary percutaneous coronary intervention: hope or hype? Heart 101:1612–1618
Nakano A, Liu GS, Heusch G, Downey JM, Cohen MV (2000) Exogenous nitric oxide can trigger a preconditioned state through a free radical mechanism, but endogenous nitric oxide is not a trigger of classical ischemic preconditioning. J Mol Cell Cardiol 32:1159–1167
Nielsen JC, Kristensen L, Andersen HR, Mortensen PT, Pedersen OL, Pedersen AK (2003) A randomized comparison of atrial and dual-chamber pacing in 177 consecutive patients with sick sinus syndrome: echocardiographic and clinical outcome. J Am Coll Cardiol 42:614–623
Obal D, Dettwiler S, Favoccia C, Scharbatke H, Preckel B, Schlack W (2005) The influence of mitochondrial KATP-channels in the cardioprotection of preconditioning and postconditioning by sevoflurane in the rat in vivo. Anesth Analg 101:1252–1260
Ovize M, Baxter GF, Di Lisa F et al (2010) Postconditioning and protection from reperfusion injury: where do we stand? Position paper from the Working Group of Cellular Biology of the Heart of the European Society of Cardiology. Cardiovasc Res 87:406–423
Penna C, Mancardi D, Raimondo S, Geuna S, Pagliaro P (2008) The paradigm of postconditioning to protect the heart. J Cell Mol Med 12:435–458
Penna C, Perrelli MG, Pagliaro P (2013) Mitochondrial pathways, permeability transition pore, and redox signaling in cardioprotection: therapeutic implications. Antioxid Redox Signal 18:556–599
Penna C, Rastaldo R, Mancardi D et al (2006) Post-conditioning induced cardioprotection requires signaling through a redox-sensitive mechanism, mitochondrial ATP-sensitive K+ channel and protein kinase C activation. Basic Res Cardiol 101:180–189
Pipicz M, Varga ZV, Kupai K et al (2015) Rapid ventricular pacing-induced postconditioning attenuates reperfusion injury: effects on peroxynitrite, RISK and SAFE pathways. Br J Pharmacol 172:3472–3483
Raedschelders K, Ansley DM, Chen DD (2012) The cellular and molecular origin of reactive oxygen species generation during myocardial ischemia and reperfusion. Pharmacol Ther 133:230–255
Rastaldo R, Cappello S, Di Stilo A, Folino A, Losano G, Pagliaro P (2012) A lipophilic nitric oxide donor and a lipophilic antioxidant compound protect rat heart against ischemia-reperfusion injury if given as hybrid molecule but not as a mixture. J Cardiovasc Pharmacol 59:241–248
Tan X, Zhang L, Jiang Y, Yang Y, Zhang W, Li Y, Zhang X (2013) Postconditioning ameliorates mitochondrial DNA damage and deletion after renal ischemic injury. Nephrol Dial Transplant 28:2754–2765
Tessier-Vetzel D, Tissier R, Waintraub X, Ghaleh B, Berdeaux A (2006) Isoflurane inhaled at the onset of reperfusion potentiates the cardioprotective effect of ischemic postconditioning through a NO-dependent mechanism. J Cardiovasc Pharmacol 47:487–492
Vanagt WY, Cornelussen RN, Baynham TC et al (2007) Pacing-induced dyssynchrony during early reperfusion reduces infarct size. J Am Coll Cardiol 49:1813–1819
Vanagt WY, Cornelussen RN, Poulina QP et al (2006) Pacing-induced dys-synchrony preconditions rabbit myocardium against ischemia/reperfusion injury. Circulation 114:I264–I269
Vinten-Johansen J, Zhao ZQ, Zatta AJ et al (2005) A new link in nature’s armor against myocardial ischemia-reperfusion injury. Basic Res Cardiol 100:295–310
Waltenberger J, Gelissen M, Bekkers SC et al (2014) Clinical pacing post-conditioning during revascularization after AMI. JACC Cardiovasc Imaging 7:620–626
Wang HC, Zhang HF, Guo WY et al (2006) Hypoxic postconditioning enhances the survival and inhibits apoptosis of cardiomyocytes following reoxygenation: role of peroxynitrite formation. Apoptosis 11:1453–1460
Wyman BT, Hunter WC, Prinzen FW, Faris OP, McVeigh ER (2002) Effects of single- and biventricular pacing on temporal and spatial dynamics of ventricular contraction. Am J Physiol Heart Circ Physiol 282:H372–H379
Yang F, Xi L 2015 Postconditioning of ischemic heart by intermittent ventricular pacing at beginning of reperfusion: novel mechanisms and utilities in interventional cardiology. Am J Physiol Heart Circ Physiol ajpheart 00835 02015
Yang XM, Krieg T, Cui L, Downey JM, Cohen MV (2004) NECA and bradykinin at reperfusion reduce infarction in rabbit hearts by signaling through PI3K, ERK, and NO. J Mol Cell Cardiol 36:411–421
Yetgin T, Magro M, Manintveld OC et al (2014) Impact of multiple balloon inflations during primary percutaneous coronary intervention on infarct size and long-term clinical outcomes in ST-segment elevation myocardial infarction: real-world postconditioning. Basic Res Cardiol 109:403
Yui H, Imaizumi U, Beppu H et al (2011) Comparative effects of verapamil, nicardipine, and nitroglycerin on myocardial ischemia/reperfusion injury. Anesthesiol Res Pract 2011:521084
Zhao ZQ, Corvera JS, Halkos ME et al (2003) Inhibition of myocardial injury by ischemic postconditioning during reperfusion: comparison with ischemic preconditioning. Am J Physiol Heart Circ Physiol 285:H579–H588
Zorov DB, Juhaszova M, Yaniv Y, Nuss HB, Wang S, Sollott SJ (2009) Regulation and pharmacology of the mitochondrial permeability transition pore. Cardiovasc Res 83:213–225
Acknowledgments
The authors want to pay special thanks to Kuwait University for the support by grant no. MY 02/10.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Babiker, F.A., Al-Jarallah, A. & Joseph, S. Understanding pacing postconditioning-mediated cardiac protection: a role of oxidative stress and a synergistic effect of adenosine. J Physiol Biochem 73, 175–185 (2017). https://doi.org/10.1007/s13105-016-0535-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13105-016-0535-z