Abstract
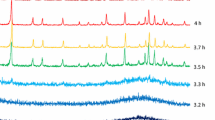
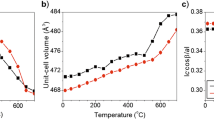
1H MAS-NMR spectroscopy reveals analogies between partly dehydrated samples of silica-alumina and strongly dealuminated HY zeolite. There is a fundamental difference between the dehydration processes of crystalline and amorphous samples. A scheme for the reactions occurring during dehydration and dehydroxylation is proposed.
Similar content being viewed by others
References
M. Hunger, D. Freude, H. Pfeifer, H. Bremer, M. Jank and K.-P. Wenlandt, Chem. Phys. Lett. 100 (1983) 29.
H. Pfeifer, D. Freude and M. Hunger, Zeolites 5 (1985) 274;
see also: J.M. Thomas and J. Klinowski, Adv. Catalysis 33 (1985) 199.
H. Pfeifer, J. Chem. Soc., Faraday Trans. I, 84 (1988) 3777.
D. Freude, Stud. Surf. Sci. Catal. 52 (1989) 169.
C.E. Bronnimann, I-Ssuer Chuang, B.L. Hawkins and G.E. Maciel, J. Am. Chem. Soc. 109 (1987) 1562.
M. Hunger, D. Freude and H. Pfeifer, J. Chem. Soc., Faraday Trans. I, in press.
D. Barthomeuf, Zeolites 10 (1990) 131.
P. Batamack, C. Dorémieux-Morin, J. Fraissard and D. Freude, J. Phys. Chem., in press.
G.H. Kühl, J. Phys. Chem. Solids 38 (1977) 1259.
J.A. Rabo and G.J. Gajda, Catal. Rev.-Sci. Eng. 31 (1989-90) 385.
S. Beran, J. Phys. Chem. 85 (1981) 1956.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Dorémieux-Morin, C., Batamack, P., Martin, C. et al. Comparison of amorphous silica-alumina and highly dealuminated HY zeolite by1H high resolution MAS-NMR of solids. Catal Lett 9, 403–409 (1991). https://doi.org/10.1007/BF00764832
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00764832