Summary
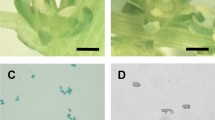
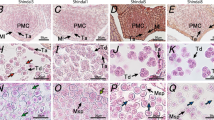
Light and fluorescence microscopy were used to study coenocytic microspore germination from male-sterile (ms1 ms1) soybean plants. Anther squashes from male-sterile plants revealed that a low frequency of natural coenocytic microspore germination occurred in male-sterile anthers of four independent lines; [ms1-North carolina (T260H),ms1-Urbana (T266H),ms1-Tonica (T267H), andms1-Ames (T268H)]. Abnormalities such as giant tubes, branched tubes, tubes with swollen areas, and multiple tubes were observed from coenocytic microspores from all four lines. The Urbana line, however, demonstrated a higher percentage of coenocytic microspore germination than did the other three lines. Flowers of the Urbana line from both malefertile and male-sterile plants, as well as gynoecia pollinated with coenocytic microspores from sterile plants, were used for in vivo studies. Pollen-tube growth appeared normal in male-fertile plants. In contrast, coenocytic microspore tubes rarely were observed in gynoecia from male-sterile plants or in gynoecia from malefertile plants that had been artificially cross-pollinated withms1 ms1 plants. Few tubes from coenocytic microspores were observed in the vicinity of the micropylar region. A low frequency of seed set was achieved in the greenhouse on Urbana male-sterile plants grown in the absence of male-fertile plants. Thus, we believe either that some gametes from coenocytic microspores are able to participate in fertilization at low frequency or that apomixis occurs inms1 ms1 plants.
Similar content being viewed by others
References
Albertsen, M. C. & R. G. Palmer, 1977. Proposed mechanism(s) for self-fertilization in male-sterile, female-fertile (ms1) soybeans (Glycine max L.Merr.). Bot. Soc. Am. Abstr. p3.
AlbertsenM. C. & R. G.Palmer, 1979. A comparative light- and electron-microscopic study of microsporogenesis in male-sterile (ms1) and male-fertile soybean (Glycine max L.Merr.). Am. J. Bot. 66: 253–265.
BerlynG. P. & J. P.Miksche, 1976. Botanical microtechnique and cytochemistry. The Iowa State University Press, Ames.
BeversdorfW. D. & E. T.Bingham, 1977. Male-sterility as a source of haploids and polyploids ofGlycine max. Can. J. Genet. Cytol. 19: 283–287.
BoermaH. R. & R. L.Cooper, 1978. Increased female fertility associated with thems1 locus in soybeans. Crop Sci. 18: 344–346.
BrewbakerJ. L. & B. H.Kwack, 1963. The essential role of calcium ion in pollen germination and pollen-tube growth. Am. J. Bot. 50: 859–865.
BrimC. A. & M. F.Young, 1971. Inheritance of a male-sterile character in soybeans. Crop Sci. 11: 564–566.
CarlsonJ. B., 1973. Morphology. p. 17–95. In: B. E.Caldwell (Ed.), Soybeans: Improvement, production, and uses. Am. Soc. Agron., Madison, Wisconsin.
ChenL. F. O., H. E.Heer & R. G.Palmer, 1985. The frequency of polyembryonic seedlings and polyploids fromms1 soybean. Theor. Appl. Genet. 69: 271–277.
Cutter, G. L., 1975. Effect of a genetic male-sterile character on the organization and function of the female gametophyte in soybeans. Ph.D. Diss. University of Wisconsin, Madison.
CutterG. L. & E. T.Bingham, 1977. Effect of soybean male-sterile genems1 on organization and function of the female gametophyte. Crop Sci. 17: 760–764.
FehrW. R., 1980. Soybean. p. 589–599. In: W. R.Fehr & H. H.Hadley (Eds), Hybridization of crop plants. Am. Soc. Agron, Madison, Wisconsin.
FehrW. R. & C. E.Caviness, 1977. Stages of soybean development. Iowa Agric. Home Econ. Exp. Stn., Iowa Coop. Ext. Serv. Spec. Rep. 80: 1–12.
GrayboschR. A. & R. G.Palmer, 1985. Male sterility in soybean (Glycine max). II. Phenotypic expression of thems4 mutant. Am. J. Bot. 72: 1751–1764.
Heslop-HarrisonJ., 1957. The experimental modification of sex expression in flowering plants. Biol. Rev. (Camb.) 32: 38–90.
JensenW. A., 1962. Botanical histochemistry. W. H. Freeman and Company, San Francisco.
KennellJ. C. & H. T.Horner, 1985a. Influence of the soybean male-sterile gene (ms1) on the development of the female gametophyte. Can. J. Genet. Cytol. 27: 200–209.
KennellJ. C. & H. T.Horner, 1985b. Megasporogenesis and megagametogenesis in soybean,Glycine max (L.)Merr. Am. J. Bot. 72: 1553–1564.
KenworthyW. J., C. A.Brim & E. A.Wernsman, 1973. Polyembryony in soybeans. Crop Sci. 13: 637–639.
KhoY. O. & J.Baer, 1968. Observing pollen tubes by means of fluorescence. Euphytica 17: 298–302.
PalmerR. G. & H.Heer, 1973. A root tip squash technique for soybean chromosomes. Crop Sci. 13: 389–391.
PalmerR. G. & H.Heer, 1976. Polyploids among progeny from male-sterile (ms1 ms1) soybean. Soybean Genet. Newsl. 3: 35–36.
PalmerR. G., C. L.Winger & M. C.Albertsen, 1978a. Four independent mutations at thems1 locus in soybeans. Crop Sci. 18: 727–729.
PalmerR. G., M. C.Albertsen & H.Heer, 1978b. Pollen production in soybeans with respect to genotype, environment, and stamen position. Euphytica 27: 427–433.
PrakashN. & Y. Y.Chan, 1976. Embryology ofGlycine max. Phytomorphology 26: 302–309.
SkorupskaH. & J.Nawracala, 1980. Observation of pollen grains of soybean plants in male-sterile line Urbanams1. Genet. Pol. 21: 63–67.
TiltonV. R., L. W.Wilcox, R. G.Palmer & M. C.Albertsen, 1974. Stigma, style, and obturator of soybean,Glycine max (L.)Merr. (Leguminosae) and their function in the reproductive process. Am. J. Bot. 71: 676–686.
Author information
Authors and Affiliations
Additional information
Joint contribution: Agricultural Research Service, U.S. Department of Agriculture, and Journal Paper No. J-12310 of the Iowa Agriculture and Home Economics Experiment Station, Ames IA, 50011 USA. Project 2471.
Rights and permissions
About this article
Cite this article
Chen, L.F., Albertsen, M.C. & Palmer, R.G. Pollen and coenocytic microspore germination in male-fertile and malesterilemsl soybean. Euphytica 36, 333–343 (1987). https://doi.org/10.1007/BF00730680
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00730680