Abstract
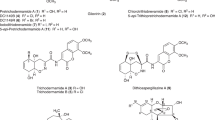
Nivalenol is a toxic trichothecene metabolite which is produced by a number of differentFusarium species. However, the nature of its immediate biosynthetic precursor is not known. Oxidation of 7-deoxynivalenol(3α,4β, 15-trihydroxy-12,13-epoxytrichothec-9-ene-8-one) to nivalenol occurred with reagents known to react by a free radical pathway, such as hydrogen peroxide-ferrous ion-ascorbic acid or lead tetracetate, but not with electrophilic reagents requiring prior formation of the enol. These results suggest that 7-deoxynivalenol or an acetylated derivative could be the biosynthetic precursor of nivalenol.
Similar content being viewed by others
References
Tatsuno T, Fujimoto Y, Morita Y. Toxicological research on substances fromFusarium nivale. The structure of nivalenol and its monoacetate, Tetrahedron Lett. 1969; 2823–2826.
Pathre SV, Mirocha CJ. Trichothecenes: natural occurrence and potential hazard: J Am Oil Chem Soc 1976; 56: 820–823.
Golinski P, Vesonder RF, Latus-Zietkiewicz D, Perkowski J. Formation of fusarenone X, nivalenol, zearalenone,α-trans-zearalenol,β-trans-zearalenol, and fusarin C byFusarium crookwellense. Appl envir Microbiol 1988; 54: 2147–2148.
Jemmali M, Ueno Y, Ishii K, Frayssinet C, Etienne M. Natural occurrence of trichothecenes (nivalenol, deoxynivalenol, T-2) and zearalenone in corn. Experientia 1978; 34: 1333–1334.
Smalley EB, Strong FM. Toxic trichothecenes inMycotoxins, IFH Purchase, Ed. Elsevier NY 1974; 199–228.
Vesonder RF, Ciegler A, Jensen AH, Rohwedder WK, Weisleder D. Co-identity of the refusal and emetic principle from Fusarium-infected corn, Appl envir Microbiol 1976; 31:280–285.
Bamburg JR, Riggs NV, Strong FM. The structures of toxins from two strains ofFusarium tricinctum, Tetrahedron 1968; 24: 3329–3336.
Ehrlich KC, Daigle KW. Protein synthesis inhibition by 8-oxo-12,13-epoxytrichothecenes. Biochim Biophys Acta 1987; 923: 206–213.
Sigg HP, Mauli R, Flury E, Hauser D. The constitution of diacetoxyscirpenol. Helv Chim Acta 1965; 48: 962–988.
Hassner A, Reuss RH, Pinnick HW. Hydroxylation of carbonyl compounds via silyl enol ethers, J Org Chem 1975; 40: 3427–3429.
Hudrlik PF, Takacs JM. Reactions of ketones with sodium hydride or potassium hydride in the presence of trimethyl silylchloride. Preparation of trimethylsilyl enol ethers. J Org Chem 1978; 43: 3861–3865.
Vedejs E, Engler DA, Telschew JE. Transition-metal peroxide reactions, synthesis ofα-hydroxycarbonyl compounds from enolates., J Org Chem 1978; 43: 188–196.
Udenfriend S, Clark CT, Axelrod J, Brodie BB. Ascorbic acid in aromatic hydroxylation. I. A model system for aromatic hydroxylation, J Biol Chem 1954; 208: 731–739.
Grove JF. Phytotoxic compounds produced byFusarium equisetii. Transformation products of 4β,15-diacetoxy-3, 7-dihydroxy-12,13-epoxytrichothec-9-ene-8-one and the structures of nivalenol and fusarenone, J Chem Soc (C) 1970; 375–378.
Zamir LO. Biosynthesis of 3-acetyldeoxynivalenol, in Steyn, PS and Vleggaar, R (Eds.),Mycotoxins and Phycotoxins, Elsevier Science Publisher B.V. 1986; 109–123.
Walling C:Free Radicals in Solution, Wiley, 70, 1957.
Norman ROC, Radda GK: Aromatic hydroxylation: the electrophilic character of the hydroxyl radical and its significance in biological hydroxylation. J Chem Soc 1962 (Proceedings): 138.
Tamm C, Breitenstein W. The biosynthesis of trichothecene mycotoxins inThe Biosynthesis of Mycotoxins, a Study in Secondary Metabolism, ed. Steyn, PS Academic Press 1980; 69–104.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Ehrlich, K.C. Preparation of theFusarium toxin, nivalenol, by oxidation of the putative biosynthetic precursor, 7-deoxynivalenol. Mycopathologia 107, 111–114 (1989). https://doi.org/10.1007/BF00707546
Issue Date:
DOI: https://doi.org/10.1007/BF00707546