Summary
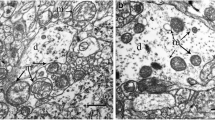
Adult normothermic cats were submitted to 1- h complete cerebrocirculatory arrest, followed by blood recirculation for 6–8 h. Two groups of animals could be distinguished: In one group electrocorticogram and somatically evoked primary cortical potentials steadily recovered after ischemia, and in another electrophysiologic recovery was absent. At the end of the recirculation period, calcium content was measured in tissue samples taken from cerebral cortex and hippocampus, and compared with mitochondrial calcium sequestration as assessed by electron-microscopic cytochemistry. Protein content of cortex and hippocampus was also determined for evaluation of tissue swelling. The two regions were selected because previous experiments had revealed that in animals with electrophysiologic recovery cerebral cortex remains intact although hippocampus is selectively injured, whereas in animals without electrophysiologic recovery both cerebral cortex and hippocampus are damaged.
In animals with functional recovery, neither calcium content nor mitochondrial calcium sequestration were significantly increased in either cerebral cortex or hippocampal subfield CA1. Only in dentate gyrus a minor degree of mitochondrial calcium sequestration was present. Calculation of tissue swelling revealed no change in cerebral cortex, but a volume increase by 18% in hippocampus, indicating development of brain edema in this region. In animals without functional recovery tissue calcium significantly increased both in cortex and hippocampus (by 49% and 73% of control, respectively), and there was significant mitochondrial calcium accumulation in both regions. Calculated brain swelling in these animals amounted to 16% and 26% in cortex and hippocampus, respectively.
The results obtained do not support the hypothesis that selective vulnerability of hippocampus is the consequence of neuronal calcium overload but rather indicate that calcium accumulation is an unspecific epiphenomenon of irreversible cell injury.
Similar content being viewed by others
References
Ames A III, Nesbett FB (1983) Pathophysiology of ischemic cell death. III. Role of extracellular factors. Stroke 14:233–240
Bodsch W, Takahashi K (1985) Selective vulnerability of hippocampal neurons to in vivo protein synthesis and protein phosphorylation. Nature (in press)
Borgers M, Thone F, Van Neuten JM (1981) The subcellular distribution of calcium and the effects of calcium-antagonists as evaluated with a combined oxalate-pyroantimonate technique. Acta Histochem [Suppl] 24:327–332
Brown AW, Brierley JB (1972) Anoxic-ischaemic cell change in rat brain. Light-microscopic and fine-structural observations. J Neurol Sci 16:59–84
Dean JM, Hoehner PJ, Rogers MC, Traystman RJ (1984) Effect of lidoflazine on cerebral blood flow following 12-min total cerebral ischemia. Stroke 15:531–535
Dienel GA (1984) Regional accumulation of calcium in postischemic rat brain. J Neurochem 43:913–925
Dienel GA, Pulsinelli WA (1984) Radioactive tracers to label selectively ischemic damaged brain. Ann Neurol 16:115–116
Farber JL, Chien KR, Mittnacht S Jr (1981) The pathogenesis of irreversible cell injury in ischemia. Am J Pathol 102:271–281
Hansen AJ, Zeuthen T (1981) Extracellular ion concentrations during spreading depression and ischemia in the rat brain cortex. Acta Physiol Scand 113:437–455
Hillered L, Siesjö BK, Arfors K-E (1984) Mitochondrial response to transient forebrain ischemia and recirculation in the rat. J Cereb Blood Flow Metabol 4:438–446
Hossmann K-A, Olsson Y (1971) The effect of transient cerebral ischemia on the vascular permeability to protein tracers. Acta Neuropathol (Berl) 18:103–112
Hossmann K-A, Paschen W, Csiba L (1983) Relationship between calcium accumulation and recovery of cat brain after prolonged cerebral ischemia. J Cereb Blood Flow Metabol 3:346–353
Hossmann K-A, Sakaki S, Kimoto K (1976a) Cerebral uptake of glucose and oxygen in the cat brain after prolonged ischemia. Stroke 7:301–305
Hossmann K-A, Sakaki S, Zimmermann V (1977) Cation activities in reversible ischemia of the cat brain. Stroke 8:77–81
Hossmann K-A, Takagi S, Sakaki S (1976b) Vital microscopy of pial arteries after prolonged cerebral ischemia. Arzneimittel Forsch 26:1233–1234
Kazda S, Towart R (1982) Nimodipine: a new calcium antagonistic drug with a preferential cerebrovascular action. Acta Neurochir 63:259–265
Kirino T (1982) Delayed neuronal death in the gerbil hippocampus following ischemia. Brain Res 239:57–69
Kleihues P, Hossmann K-A, Pegg AE, Kobayashi K, Zimmermann V (1975) Resuscitation of the monkey brain after 1-h complete ischemia. III. Indications of metabolic recovery. Brain Res 95:61–73
Korf J, Postema F (1984) Regional calcium accumulation and cation shifts in rat brain by kainate. J Neurochem 43:1052–1060
Korf J, Zoethout FHA, Postema F (1983) Regional calcium levels in rat and mouse brain: automated fluorimetric assay and effects of centrally acting drugs. Psychopharmacology 81:275–280
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–275
Naruse S, Horikawa Y, Tanaka C, Hirakawa K, Nishikawa H, Watari H (1984) In vivo measurement of energy metabolism and the concomitant monitoring of electroencephalogram in experimental cerebral ischemia. Brain Res 296:370–372
Nicholls DG, Akerman KEO (1982) Mitochondrial calcium transport. Biochim Biophys Acta 683:57–88
Paschen W, Hossmann K-A, van den Kerckhoff W (1983) Regional assessment of energy-producing metabolism following prolonged complete ischemia of cat brain. J Cereb Blood Flow Metabol 3:321–329
Raichle ME (1983) The pathophysiology of brain ischemia. Ann Neurol 13:2–10
Ross DH, Medina MA, Cardenas HL (1974) Morphine and ethanol: Selective depletion of regional brain calcium. Science 186:63–64
Simon RP, Griffiths T, Evans MC, Swan JH, Meldrum BS (1984) Calcium overload in selectively vulnerable neurons of the hippocampus during and after ischemia: An electron microscopy study in the rat. J Cereb Blood Flow Metabol 4:350–361
Steen PA, Newberg LA, Milde JH, Michenfelder JD (1983) Nimodipine improves cerebral blood flow and neurologic recovery after complete cerebral ischemia in the dog. J Cereb Blood Flow Metabol 3:38–43
Suzuki R, Yamaguchi T, Li C-L, Klatzo I (1983) The effects of 5-min ischemia in Mongolian gerbils. II. Changes of spontaneous neuronal activity in cerebral cortex and CA1 sector of hippocampus. Acta Neuropathol (Berl) 60:217–222
White BC, Winegar CD, Jackson RE, Royce EM, Vigor DN, Hoehner TJ, Krause GS, Wilson RF (1983) Cerebral cortical perfusion during and following resuscitation from cardiac arrest in dogs. Am J Emerg Med 2:128–138
Yanagihara T, McCall JT (1982) Ionic shift in cerebral ischemia. Life Sci 30:1921–1925
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Hossmann, K.A., Ophoff, B.G., Schmidt-Kastner, R. et al. Mitochondrial calcium sequestration in cortical and hippocampal neurons after prolonged ischemia of the cat brain. Acta Neuropathol 68, 230–238 (1985). https://doi.org/10.1007/BF00690200
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00690200