Summary
-
1.
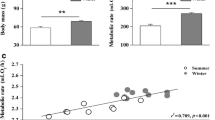
The response of the avian sympathetic nervous system during cold exposure was examined by injecting3H-norepinephrine and estimating norepinephrine (NE) turnover rate in skeletal muscle, heart, liver, and brain of seasonally acclimatized redpolls (a granivorous passerine) in interior Alaska.
-
2.
Thermoneutral NE turnover rate (ng NE/g·h) was greater in winter (January) than in summer (July) for muscle (12 ng/g·h, +115%) and heart (65 ng/g·h, +62%). This may be correlated to higher resting metabolism of winter redpolls.
-
3.
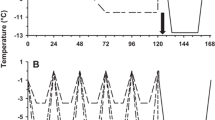
Acute cold exposure (24 h) necessary to increase metabolism 2 x caused increased NE turnover, compared to thermoneutral NE turnover, in muscle (10 ng/g·h, +95% in summer; 18 ng/g·h, +57% in winter) and heart (88 ng/g·h, +117% in summer; 108 ng/g·h, +65% in winter).
-
4.
Endogenous NE concentration declined during cold exposure of summer redpolls by a mean of 22% in muscle, liver, and brain, but not in winter birds.
-
5.
Thus, sympathetic nerve activity was stimulated by cold exposure in both seasonal groups, but winter acclimatization may involve an increased capacity to maintain steady state NE levels.
-
6.
A peripheral NE injection (2 mg NE/kg body weight) in winter redpolls depressed metabolism by a mean of 31% from resting metabolism of 7.2 ml O2/g·h. This may be due to the effect of NE on thermoregulatory centers in the brain.
Similar content being viewed by others
Abbreviations
- NE :
-
norepinephrine
References
Allen DJ, Marley E (1967) Effect of sympathomimetic and allied amines on temperature and oxygen consumption in chickens. Br J Pharmacol Chemother 31:290–312
Beley A, Beley L, Rochette L, Bralet J (1976) Evolution in vivo of the synthesis rate of catecholamines in various peripheral organs of the rat during cold exposure. Pfluegers Arch 366:259–264
Bralet J, Beley A, Lallemant AM (1972) Modifications du taux de renouvellement de la noradrénaline dans différents organes periphériques du rat au cours de l'exposition et de l'acclimation du froid. Pfluegers Arch 335:186–197
Brodie BB, Costa E, Dlabac A, Neff NH, Smookler HH (1966) Application of steady state kinetics to the estimation of synthesis rate and turnover time of tissue catecholamines. J Pharmacol Exp Ther 154:493–498
Calder C, King JR (1974) Thermal and caloric relations of birds. In: Farner DS, King JR (eds) Avian biology IV. Academic Press, New York London, pp 259–413
Davydov AF, Shalyapina VG (1976) Influence of cold on the catecholamine content of the pectoralis major muscle in passerine birds. J Evol Biochem Physiol 12:423–425
El-Halawani ME, Waibel PE (1976) Brain indole and catecholamines of turkeys during exposure to temperature stress. Am J Physiol 230:110–115
El-Halawani ME, Wilson WO, Burger RE (1970) Cold acclimation and the role of catecholamines in body temperature regulation in male leghorns. Poultry Sci 49:621–636
El-Halawani ME, Waibel PE, Appel JR, Good AL (1973) Effects of temperature stress on catecholamines and corticosterone of male turkeys. Am J Physiol 224:384–388
Euler US von, Lishajko F (1961) Improved technique for the fluorometric estimation of catecholamines. Acta Physiol Scand 51:348–355
Feist DD (1970) Brown fat norepinephrine contents and turnover during cold acclimation and hibernation in the golden hamster (Mesocricetus auratus). Comp Gen Pharmacol 1:299–315
Feist DD (1980) Norepinephrine turnover in brown fat, skeletal muscle, and spleen of cold exposed and cold acclimated Alaskan red-backed voles. J Therm Biol 5:89–94
Feist DD, Feist CF (1978) Catecholamine synthesizing enzymes in adrenals of seasonally acclimatized voles. J Appl Physiol 44:59–62
Foster DO, Frydman ML (1979) Tissue distribution of cold-induced thermogenesis in conscience warm- and cold-acclimated rats reevaluated from changes in tissue blood flow: the dominant role of brown adipose tissue in the replacement of shivering by non shivering thermogenesis. Can J Physiol Pharmacol 57:257–270
Freeman BM (1971) Non-shivering thermogenesis in birds. In: Jansky L (ed) Non-shivering thermogenesis. Proceedings of the Symposium. Swets and Zeitlinger, Amsterdam, pp 83–96
Hart JS (1971) Rodents. In: Whittow GD (ed) Comparative physiology of thermoregulation, vol II, Mammals. New York London, Academic Press, pp 2–149
Heldmaier G (1971) Relationship between non-shivering thermogenesis and body size. In: Jansky L (ed) Non-shivering thermogenesis. Proceedings of the Symposium. Swets and Zeitlinger, Amsterdam, pp 73–81
Hillman PE, Scott NR, Tienhoven A van (1977) Impact of centrally applied biogenic amines upon the energy balance of fowl. Am J Physiol 232:R137-R144
Hissa R, Pyornila A (1977) Effect of intrahypothalamic phentolamine on hypothermia produced by peripheral noradrenaline in the pigeon. Br J Pharmacol 61:163–166
Hissa R, Rautenberg W (1974) The influence of centrally applied noradrenaline on shivering and body temperature in the pigeon. J Physiol (Lond) 238:421–435
Hissa R, Rautenberg W (1975) Thermoregulatory effects of intrahypothalamic injections of neurotransmitters and their inhibitors in the pigeon. Comp Biochem Physiol [A] 51:319–326
Hissa R, Pyornila A, Saarela S (1975 a) Effect of peripheral noradrenaline on thermoregulation in temperature acclimated pigeons. Comp Biochem Physiol [C] 51:243–247
Hissa R, Rantala K, Jeronen E (1975 b) Effect of noradrenaline on the oxygen uptake in vitro by the pigeon. Gen Pharmacol 6:303–307
Hissa R, Saarela S, Pyornila A (1975 c) Thermoregulatory effects of peripheral injections of monoamines on the pigeon. Comp Biochem Physiol [C] 51:235–241
Hissa R, George JC, Saarela S (1980) Dose-related effects of noradrenaline and corticosterone on temperature regulation in pigeon. Comp Biochem Physiol [C] 65:25–32
Hohtola E, Rintamaki H, Hissa R (1978) Differential impairment of thermogenesis in the pigeon after chemical sympathectomy. Can J Physiol Pharmacol 56:578–584
Jansky L (1973) Non-shivering thermogenesis and its thermoregulatory significance. Biol Rev 48:85–132
Johnston DW (1971) The absence of brown adipose tissue in birds. Comp Biochem Physiol [A] 40:1107–1108
Kennedy DR, Hammond RP, Hamolsky MW (1977) Thyroid and cold acclimation influences on norepinephrine metabolism in brown fat. Am J Physiol 232:E565-E569
Kopin IJ, Gordon EK (1963) Origin of norepinephrine in the heart. Nature 199:1289
Morrison PR, Grodzinsky W (1975) Morrison respirometer and determination of ADMR. In: Grodzinsky W, Klekowski RZ, Duncan A (eds) Methods for ecological bioenergetics, IBP handbook no 24. Blackwell, Oxford, pp 300–309
Oliverio A, Stjärne L (1965) Acceleration of noradrenaline turnover in the mouse heart by cold exposure. Life Sci 4:2339–2343
Quek ES, Buttery JE, DeWitt GF (1975) Catecholamines in urine: an evaluation of alumina-trihydroxyindole methods and a description of an improved method. Clin Chim Acta 58:137–144
Saarela S, Hissa R, Hohtola E, Jeronen E (1977) Effects of α-methyl-para-tyrosine and temperature stress on monoamine and metabolite levels in pigeon. J Therm Biol 2:121–126
Simmonds MA (1969) Effect of environmental temperature on the turnover of noradrenaline in hypothalamus and other areas of rat brain. J Physiol (Lond) 203:199–210
Sturkie PD (1967) Cardiovascular effects of acclimatization to heat and cold in chickens. J Appl Physiol 22:13–15
Tedesco JS, Flattery KV, Sellers EA (1977) Effects of thyroid hormones and cold exposure on turnover of norepinephrine in cardiac and skeletal muscle. Can J Physiol Pharmacol 55:515–522
West GC (1972) The effects of acclimation and acclimatization on the resting metabolic rate of the common redpoll. Comp Biochem Physiol [A] 43:293–310
Whitby LG, Axelrod J, Weil-Malherbe H (1961) The fate of3H-norepinephrine in animals. J Pharmacol Exp Ther 132: 193–201
Young JB, Landsberg L (1979) Effect of diet and cold exposure on norepinephrine turnover in pancreas and liver. Am J Physiol 236:E525-E533
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Koban, M., Feist, D.D. The effect of cold on norepinephrine turnover in tissues of seasonally acclimatized redpolls (Carduelis flammea). J Comp Physiol B 146, 137–144 (1982). https://doi.org/10.1007/BF00688727
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00688727