Summary
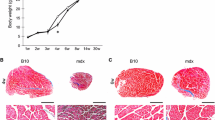
Ultrastructurally there are some clear differences in the pathology of muscle in X chromosomelinked muscular dystrophy of the mouse (mdx) and Duchenne muscular dystrophy (DMD). In particular the mouse muscle does not become infiltrated by large aggregations of connective tissue. It has been proposed that the differences are due to secondary biochemical changes consequent on the absence of dystrophin in both conditions. If this is the case, attention should be directed to the earliest ultrastructural changes held in common by both disorders. The most conspicuous of these, preceding myofibril breakdown, is dilation of the sarcoplasmic reticulum. Any physiological link between this and the absence of dystrophin remains to be determined. We suggest that in themdx mouse, the widespread myofibre necrosis occurring at 3–4 weeks is triggered by increased mechanical demands causing the lack of dystrophin to become critical at this time. Subsequent regeneration of the myofibres appears to be almost completely successful. The ultimate failure of regeneration in DMD, in contrast, may be due to an additional factors acting in DMD exacerbating the lack of dystrophin. This additional factor may be associated with the plasma membrane lesions (not seen inmdx). Alternatively there may be factors present in the mouse that compensate for the lack of dystrophin. It is pointed out that to understand better the different processes occurring inmdx and DMD we need to learn more about the factors which control the balance between the growth of muscle and the growth of connective tissue in both normal and pathological human and mouse muscle.
Similar content being viewed by others
References
Bell CD, Conen PE (1968) Histopathological changes in Duchenne muscular dystrophy. J Neurol Sci 7:529–544
Bradley WG, Fulthorpe JJ (1978) Studies of sarcolemmal integrity in myopathic muscle. Neurology 28:670–677
Bradley WG Jaros E (1979) Involvement of peripheral and central nerves in murine dystrophy. Ann NY Acad Sci 317:132–142
Bradley WG, Jenkison M (1975) Meural abnormalities in the dystrophic mouse. J Neurol Sci 72:147–157
Bridges LR (1986) The association of cardiac muscle necrosis and inflammation with the degenerative and persistent myopathy of MDX mice. J Neurol Sci 72:145–157
Bulfield G, Siller WG, Wight PAL, More K (1984) X chromosome-linked muscular dystrophy (mdx) in the mouse. Proc Natl Acad Sci USA 81:1189–1192
Carnwath JW, Shotton DM (1987) Muscular dystrophy in themdx mouse: histopathology of the soleus and extensor digitorum longus muscles. J Neurol Sci 80:39–54
Carpenter S, Karpati G (1979) Duchenne muscular dystrophy: plasma membrane loss initiates muscle cell necrosis unless it is repaired. Brain 102:147–161
Cullen MJ, Fulthorpe JJ (1982) Phagocytosis of the A band following Z line and I band loss. Its significance in skeletal muscle breakdown. J Pathol 138:129–143
Cullen MJ, Mastaglia FL (1980) Morphological changes in dystrophic muscle. Br Med Bull 36:145–152
Cullen MJ, Mastaglia FL (1982) Pathological reactions of skeletal muscle. In: Mastaglia FL, Walton JN (eds) Skeletal muscle pathology. Churchill Livingstone, Edinburgh, pp 88–139
Dangain J, Vrbova G (1984) Muscle development inmdx mutant mice. Muscle Nerve 7:700–704
Engel AG (1986) Duchenne dystrophy. In: Engel AG, Banker BQ (eds) Myology. McGraw-Hill, New York, pp 1185–1240
Engel AG, Macdonald RD (1970) Ultrastructural reactions in muscle disease and their light microscopic correlates. Excerpta Med Int Congr Ser 199:71–89
Erb WH (1891) Dystrophia muscularis progressiva: klinische und pathologisch-anatomische Studien. Dtsch Z Nervenheilkd 1:13–94
Erb WH (1891) Dystrophia muscularis progressiva: klinische und pathologisch-anatomische Studien. Dtsch Z Nervenheilkd 1:173–261
Hoffman EP, Brown RH, Kunkel LM (1987) Dystrophin: the protein product of the Duchenne muscular dystrophy locus. Cell 51:919–928
Hoffman EP, Knudson CM, Campbell KP, Kunkel LM (1987) Subcellular fractionation of dystrophin to the triads of skeletal muscle. Nature 330:754–759
Hoffman EP, Monaco AP, Feener CC, Kunkel LM (1987) Conservation of the Duchene muscular dystrophy gene in mice and humans. Science 2398:347–350
Jaros J, Johnstone D (1985) Factors affecting phenotypic expression of dystrophy in skeletal muscles of the dystrophic mouse (dy/dy). In: Ten Kate LP, Pearson PL, Stadhouders AM (eds) Research into the origin and treatment of muscular dystrophy. Excerpta Medica Current Clinical Practic, Ser No 20. Elsevier, Amsterdam, pp 103–124
Karpati G, Carpenter S (1983) Pathology of skeletal muscle. Churchill Livingstone, New York
Meltzer HY, Kund RW, Click J, Yang V (1976) Incidence of Z band streaming and myofibrillar disruptions in skeletal muscle from healthy young people. Neurology 26:853–857
Schmalbruch H (1982) The muscular dystrophies. In: Mastaglia FL, Walton JN (eds) Skeletal muscle pathology. Churchill Livingstone, Edinburgh, 235–265
Schmalburch H (1984) Regenerated muscle fibers in Duchenne muscular dystrophy: a serial section study. Neurology 34:60–65
Somlyo AV, Gonzalez-Serratos H, Shuman H, McClellan G, Somlyo AP (1981) Calcium release and ionic changes in the sarcoplasmic reticulum of tetanized muscle — an electron-probe study. J Cell Biol 90:577–594
Tanabe Y, Esaki K, Nomura T (1986) Skeletal muscle pathology in X chromosome-linked muscular dystrophy (mdx) mouse. Acta Neuropathol (Berl) 69:91–95
Torres LFB, Duchen LW (1987) The mutantmdx: inherited myopathy in the mouse: morphological studies of nerves, muscles and endpaltes. Brain 110:269–300
Watkins SC, Cullen MJ (1985) Histochemical fibre typing and ultrastructure of the small fibres in Duchenne muscular dystrophy. Neuropathol Appl Neurobiol 11:447–460
Watkins SC, Cullen MJ (1987) A qualitative and quantitative study of muscle fibre regeneration in Duchenne muscular dystrophy and polymyositis. J Neurol Sci 82:181–192
Author information
Authors and Affiliations
Additional information
Supported by the Muscular Dystrophy Group of Great Britain and the Medical Research Council
Rights and permissions
About this article
Cite this article
Cullen, M.J., Jaros, E. Ultrastructure of the skeletal muscle in the X chromosome-linked dystrophic (mdx) mouse. Acta Neuropathol 77, 69–81 (1988). https://doi.org/10.1007/BF00688245
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00688245