Abstract
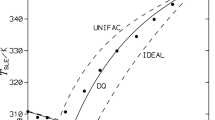
Freezing point temperatures for binary mixtures containing phenylazophenol (PAP) with p-(p-methoxyphenylazo)phenol (MOPAP), p-(p-ethylphenylazo)phenol (EPAP) and p-(p-ethoxyphenylazo)phenol (EOPAP) have been experimentally determined using differential scanning calorimetry. Enthalpies and entropies of fusion for the four phenylazophenol derivatives were also measured. Results of these measurements indicate that the three binary systems exhibit only slight deviations from ideality at temperatures between the melting points of the pure components. Deviations become more pronounced in the PAP+EOPAP system as the temperature decreases below the melting pont of the ethoxy-derivative.
Similar content being viewed by others
References
G. W. Gray and P. A. Winsor,Liquid Crystals and Plastic Crystals (Wiley, New York, 1974).
W. E. Acree, Jr.,Thermodynamic Properties of Nonelectrolyte Solutions (Academic Press, Orlando, FL, 1984), Chap. 10.
J. M. Prausnitz, R. N. Lichtenthaler, and E. Gomes de Azevedo,Molecular Thermodynamics of Fluid Phase Equilibria, 2nd edn. (Prentice-Hall, Englewood Cliffs, NJ, 1986), Chap. 9.
Y. Matsunaga,Bull. Chem. Soc. Jap. 29, 308 (1956).
G. L. Eian and C. A. Kingsbury,Bull. Chem. Soc. Jap. 43, 739 (1970).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Djordjevic, N.M., Rouse, C.A., Djerki, R.A. et al. Solubility ofp-phenylazophenol in low molecular weight liquid crystalline precursors. J Solution Chem 17, 967–975 (1988). https://doi.org/10.1007/BF00649740
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00649740