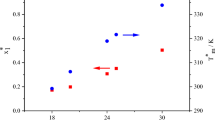
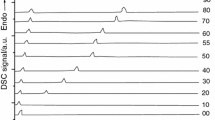
Abstract
A differential scanning calorimetric technique has been used to obtain solid–liquid equilibrium temperatures for the mixtures naphthalene or biphenyl + 1-tetradecanol, or + 1-hexadecanol. All the systems show a simple eutectic point, whose final composition was determined by means of the Tamman’s plots using the needed values of the eutectic heat and of the heat of melting, which are also reported. DISQUAC interaction parameters for the OH/aromatic contacts in the selected systems are given. The present experimental SLE phase diagrams are similarly described by DISQUAC and UNIFAC (Dortmund) models. However, the comparison of DISQUAC and UNIFAC results for systems involving naphthalene and shorter 1-alkanols (methanol + 1-octanol) reveals that the temperature dependence of the interaction parameters is more suitable in DISQUAC. The systems are also investigated in terms of the concentration-concentration structure factor. It is shown that the positive deviations from the Raoult’s law of the studied solutions become weaker when the homocoordination decreases.









Similar content being viewed by others
References
Farahani, B.V., Rajabi, F.H., Hosseindoust, B., Zenooz, N.: DSC study of solid–liquid equilibria for energetic binary mixtures of methylnitramine with 2,4-dinitro-2,4-diazapentane and 2,4-dinitro-2,4-diazahexane. J. Phase Equilib. Diffus. 31, 536–541 (2010)
Ulrich, J., Bülau, H.C.: Melt crystallization. In: Myerson, A.S. (ed.) Handbook of Industrial Crystallization, pp. 161–179. Butterworth-Heinemann, Oxford (2002)
Ahlers, J., Lohmann, J., Gmehling, J.: Binary solid–liquid equilibria of organic systems containing different amides and sulfolane. J. Chem. Eng. Data 44, 727–730 (1999)
Wittig, R., Constantinescu, D., Gmehling, J.: Binary solid–liquid equilibria of organic systems containing-caprolactone. J. Chem. Eng. Data 46, 1490–1493 (2001)
Sarbu, I., Sebarchievici, C.A.: Comprehensive review of thermal energy storage. Sustainability 10, 191 (2018)
Fallahi, A., Guldentops, G., Tao, M., Granados-Focil, S., Van Dessel, S.: Review on solid–solid phase change materials for thermal energy storage: molecular structure and thermal properties. Appl. Therm. Eng. 127, 1427–1441 (2017)
Abbott, A.P., Boothby, D., Capper, G., Davies, D.L., Rasheed, R.K.: Deep eutectic solvents formed between choline chloride and carboxylic acids: versatile alternatives to ionic liquids. J. Am. Chem. Soc. 126, 9142–9147 (2004)
Abbott, A.P., Capper, G., Davies, D.L., Rasheed, R.K., Tambyrajah, V.: Novel solvent properties of choline chloride/urea mixtures. Chem. Commun. (2003). https://doi.org/10.1039/b210714g
Foreman, M.R.S.: Progress towards a process for the recycling of nickel metal hydride electric cells using a deep eutectic solvent. Cogent Chem. 2, 1139289–1139300 (2016)
Łukomska, A., Wiśniewska, A., Dąbrowski, Z., Kolasa, D., Luchcińska, S., Lach, J., Wróbel, K.: Recovery of zinc and manganese from “black mass” of waste Zn–MnO2 alkaline batteries by solvent extraction technique with ionic liquids, DES and organophosphorous-based acids. J. Mol. Liq. 338, 116590 (2021)
Domanska, U.: Solubility and hydrogen bonding. Part VII. Synergic effect of solubility of naphthalene in mixed solvents. Polish. J. Chem. 55, 1715–1720 (1981)
Ward, H.L.: The solubility relations of naphthalene. J. Phys. Chem. 30, 1316–1333 (1926)
Boudouh, I., González, J.A., Djemani, I., Barkat, D.: Solid-liquid equilibria of eicosane, tetracosane, or biphenyl + 1-octadecanol, or + 1-eicosanol mixtures. Fluid Phase Equilib. 442, 28–37 (2017)
Hawes, D.W., Feldman, D., Banu, D.: Latent heat storage in building materials. Energy Build. 20, 77–86 (1993)
Burley, S.K., Petsko, G.A.: Weakly polar interactions in proteins. Adv. Protein Chem. 39, 125–189 (1988)
Cabaleiro, D., Gracia-Fernández, C., Lugo, L.: (Solid–liquid) phase equilibria and heat capacity of (diphenyl ether + biphenyl) mixtures used as thermal energy storage materials. J. Chem. Thermodyn. 74, 43–50 (2014)
Cabaleiro, D., Pastoriza-Gallego, M.J., Piñeiro, M.M., Legido, J.L., Lugo, L.: Thermophysical properties of (diphenyl ether + biphenyl) mixtures for their use as heat transfer fluids. J. Chem. Thermodyn. 50, 80–88 (2012)
Demirbas, A.: Asphaltene yields from five types of fuels via different methods. Energy Convers. Manag. 43, 1091–1097 (2002)
Silva, S.M.C., Rajagopal, K.: Steady state size distribution of asphaltenes by flocculation from toluene-n-heptane mixtures. Petrol. Sci. Technol. 22, 1073–1085 (2004)
Wilson, G.M.: Vapor–liquid equilibrium. XI. A new expression for the excess free energy of mixing. J. Am. Chem. Soc. 86, 127–130 (1964)
Renon, H., Prausnitz, J.M.: Local compositions in thermodynamic excess functions for liquid mixtures. AIChE J. 14, 135–144 (1968)
Weidlich, U., Gmehling, J.: A modified UNIFAC model. 1. Prediction of VLE, HE and γ∞. Ind. Eng. Chem. Res. 26, 1372–1381 (1987)
Kehiaian, H.V.: Group contribution methods for liquid mixtures: a critical review. Fluid Phase Equilib. 13, 243–252 (1983)
González, J.A., García de la Fuente, I., Cobos, J.C.: Correlation and prediction of excess molar enthalpies using DISQUAC. In: Wilhelm, E., Letcher, T.M. (eds.) Enthalpy and Internal Energy Liquids, Solutions and Vapours. Royal Society of Chemistry, Croydon (2017)
Gmehling, J., Li, J., Schiller, M.: A modified UNIFAC model, 2. Present parameter matrix and results for different thermodynamic properties. Ind. Eng. Chem. Res. 32, 178–193 (1993)
Gonzalez, J.A., Zawadzki, M., Domanska, U.: Thermodynamics of mixtures containing polycyclic aromatic hydrocarbons. J. Mol. Liq. 143, 134–140 (2008)
Domanska, U., González, J.A.: Solid–liquid equilibria for systems containing long-chain 1-alkanols. I. Experimental data for 1-dodecanol, 1-tetradecanol, 1-hexadecanol, 1-octadecanol or 1-icosanol + benzene, or toluene mixtures. Characterization in terms of DISQUAC. Fluid Phase Equilib. 119, 131–151 (1996)
Cobos, J.C.: An exact quasi-chemical equation for excess heat capacity with W-shaped concentration dependence. Fluid Phase Equilib. 133, 105–127 (1997)
Trejo, L.M., Costas, M., Patterson, D.: Effect of molecular size on the W-shaped excess heat capacities: oxaalkane–alkane systems. J. Chem. Soc. Faraday Trans. 87, 3001–3008 (1991)
CIAAW, Atomic weights of the elements 2015, ciaaw.org/atomic-weights.htm (2015).
Stolen, S., Gronvold, F.: Critical assessment of the enthalpy of fusion of metals used as enthalpy standards at moderate to high temperatures. Thermochim. Acta 327, 1–32 (1999)
Mosselman, C., Mourik, J., Dekker, H.: Enthalpies of phase change and heat capacities of some long-chain alcohols. Adiabatic semi-microcalorimeter for studies of polymorphism. J. Chem. Thermodyn. 6, 477–487 (1974)
Metivaud, V., Lefevre, A., Ventola, L., Negrier, P., Moreno, E., Calvet, T., Mondieig, D., Cuevas-Diarte, M.A.: Hexadecane (C16H34) + 1-hexadecanol (C16H33OH) binary system: crystal structures of the components and experimental phase diagram. Application to thermal protection of liquids. Chem. Mater. 17, 3302–3310 (2005)
Kuchhal, Y.K., Shukla, R.N., Biswas, A.B.: Differential thermal-analysis of n-long chain alcohols and corresponding alkoxy ethanols. Thermochim. Acta 31, 61–70 (1979)
Berchiesi, G.: Data mixtures. Int. DATA Ser. Selec. Ser. A 2, 95–100 (1985)
Prausnitz, J.M., Lichtenthaler, R.N.: Termodinámica Molecular de los Equilibrios Entre Fases. Prentice-Hall, Madrid (2000)
Boudouh, I., Hafsaoui, S.L., Mahmoud, R., Barkat, D.: Measurement and prediction of solid-phase equilibria for systems containing biphenyl in binary solution with long-chain n-alkanes. J. Therm. Anal. Calorim. 125, 793–801 (2016)
Costa, M.C., Rolemberg, M.-P., Meirelles, A.J.A., Coutinho, J.P.A., Krähenbühl, M.A.: The solid–liquid phase diagrams of binary mixtures of even saturated fatty acids differeing by six carbon atoms. Thermochim. Acta 496, 30–37 (2009)
Inoue, T., Hisatsugu, Y., Ishikawa, R., Suzuki, M.: Solid–liquid phase behaviour of binary fatty acid mixtures: 2. Mixtures of oleic acid with lauric acid, myristic acid, and palmitic acid. Chem. Phys. Lipids 127, 161–173 (2004)
Grzyll, L.R., Ramos, C., Back, D.D.: Density, viscosity, and surface tension of liquid quinoline, naphthalene, biphenyl, decafluorobiphenyl, and 1,2-diphenylbenzene from 300 to 400 °C. J. Chem. Eng. Data 41, 446–450 (1996)
Liew, K.Y., Seng, C.E., Ng, B.H.: Molar volumes of n-alcohols from 15 to 80 °C. J. Solution Chem. 21, 1177–1183 (1992)
Guggenheim, E.A.: Mixtures. Oxford University Press, Oxford (1952)
Bondi, A.: Physical Properties of Molecular Crystals, Liquids and Glasses. Wiley, New York (1968)
Kehiaian, H.V., Grolier, J.-P.E., Benson, G.C.: Thermodynamics of organic mixtures. A generalized quasichemical model theory in terms of group surface interactions. J. Chim. Phys. 75, 1031–1048 (1978)
González, J.A., García de la Fuente, I., Cobos, J.C., Casanova, C., Ait-Kaci, A.: Application of the zeroth approximation of the DISQUAC model to cyclohexane + n-alkane mixtures using different combinatorial entropy terms. Fluid Phase Equilib. 112, 63–87 (1995)
González, J.A., García de la Fuente, I., Cobos, J.C.: Thermodynamics of mixtures with strongly negative deviations from Raoult’s law. Part 4. Application of the DISQUAC model to mixtures of 1-alkanols with primary or secondary linear amines. Comparison with Dortmund UNIFAC and ERAS results. Fluid Phase Equilib. 168, 31–58 (2000)
Fredenslund, A., Jones, R.L., Prausnitz, J.M.: Group-contribution estimation of activity coefficients in nonidel liquid mixtures. AIChE J. 21, 1086–1099 (1975)
Bevington, P.R.: Data Reduction and Error Analysis for the Physical Sciences. McGraw-Hill, New York (1969)
Boudouh, I., Djemai, I., González, J.A., Barkat, D.: Solid–liquid equilibria of biphenyl binary systems. J. Mol. Liq. 216, 764–770 (2016)
González, J.A., García de la Fuente, I., Cobos, J.C., Casanova, C.: A characterization of the aliphatic/hydroxyl interactions using a group contribution model (DISQUAC). Ber. Bunsenges. Phys. Chem. 95, 1658–1668 (1991)
González, J.A., García de la Fuente, I., Cobos, J.C., Casanova, C., Domanska, U.: DISQUAC application to SLE of binary mixtures containing long chain 1-alkanols (1-tetradecanol, 1-hexadecanol, 1-octadecanol, or 1-eicosanol) and n-alkanes (C8–C16). Ber. Bunsenges. Phys. Chem. 98, 955–959 (1994)
Kang, J.W., Diky, V., Frenkel, M.: New modified UNIFAC parameters using critically evaluated phase equilibrium data. Fluid Phase Equilib. 388, 128–141 (2015)
Ott, J.B., Holscher, I.F., Schneider, G.M.: (Liquid + liquid) phase equilibria in (methanol + heptane) and (methanol + octane) at pressures from 0.1 to 150 MPa. J. Chem. Thermodyn. 18, 815–826 (1986)
Aoulmi, A., Bouroukba, M., Solimando, R., Rogalski, M.: Thermodynamics of mixtures formed by polycyclic aromatic hydrocarbons with long chain alkanes. Fluid Phase Equilib. 110, 283–297 (1995)
Coon, J.E., Sediawan, W.B., Auwaerter, J.E., McLaughlin, E.: Solubilities of families of heterocyclic polynuclear aromatics in organic solvents and their mixtures. J. Solution Chem. 17, 519–534 (1988)
Chirico, R.D., Knipmeyer, S.E., Nguyen, A., Steele, W.V.: The thermodynamic properties of biphenyl. J. Chem. Thermodyn. 21, 1307–1331 (1989)
Leys, J., Losada-Pérez, P., Slenders, E., Glorieux, C., Thoen, J.: Investigation of the melting behaviour of the reference materials biphenyl and phenyl salicytate by a new type adiabatic scanning calorimeter. Thermochim. Acta 582, 68–76 (2014)
Sharma, K.P., Rai, R.N.: Synthesis and characterization of novel binary organic monotectic and eutectic alloys. Thermochim. Acta 535, 66–70 (2012)
Chirico, R.D., Knipmeyer, S.E., Steele, W.V.: Heat capacities, enthalpy increments, and derived thermodynamic functions for naphthalene between the temperatures 5 K and 440 K. J. Chem. Thermodyn. 34, 1874–1884 (2002)
Khimeche, K., Dahmani, A.: Solid–liquid equilibria of naphthalene + alkanediamine mixtures. J. Chem. Eng. Data 51, 383–385 (2006)
Acree, W.E.: Thermodynamic properties of organic compounds: enthalpy of fusion and melting point temperature compilation. Thermochim Acta 189, 37–56 (1991)
Sharma, B.L., Gupta, S., Tandon, S., Kant, R.: Physico-mechanical properties of naphthalene–acenaphthene eutectic system by different modes of solidification. Mater. Chem. Phys. 111, 423–430 (2008)
Maximo, G.J., Carareto, N.D.D., Costa, M.C., Dos, S.: On the solid–liquid equilibrium of binary mixtures of fatty alcohols and fatty acids. Fluid Phase Equilib. 366, 88–98 (2014)
Tian, T., Song, J., Niu, L., Feng, R.: Preparation and properties of 1-tetradecanol/1,3:2,4-di-(3,4-dimethyl) benzylidene sorbitol gelatinous form-stable phase change materials. Thermochim. Acta 554, 54–58 (2013)
Zuo, J., Li, W., Weng, L.: Thermal properties of lauric acid/1-tetradecanol binary system for energy storage. App. Thermal Eng. 31, 1352–1355 (2011)
Davies, M., Kybett, B.: Sublimation and vaporization heats of long-chain alcohols. Trans. Faraday Soc. 61, 1608–1617 (1965)
Xing, J., Tan, Z.-C., Shi, Q., Tong, B., Wang, S.-X., Li, Y.-S.: Heat capacity and thermodynamic properties of 1-hexadecanol. J. Themal Anal. Calorim. 92, 375–380 (2008)
Wiśniewska, B., Gregorowicz, J., Malanowski, S.: Development of a vapour–liquid equilibrium apparatus to work at pressures up to 3 MPa. Fluid Phase Equilib. 86, 173–186 (1993)
Lee, C.H., Holder, G.D.: Vapor–liquid equilibria in the systems toluene/naphthalene and cyclohexane/naphthalene. J. Chem. Eng. Data 38, 320–323 (1993)
Butcher, K.L., Medani, M.S.: Thermodynamic properties of methanol–benzene mixtures at elevated temperatures. J. Appl. Chem. 18, 100–107 (1968)
Funding
This work was supported by Consejería de Educación de Castilla y León, under Project VA100G19 (Apoyo a GIR, BDNS: 425389.
Author information
Authors and Affiliations
Contributions
L.F. Sanz and F. Hevia performed the experimental work. Data correlation was conducted by J.A. González and L.F. Sanz. J.A. González, I. García de la Fuente and J.C. Cobos wrote the draft of the manuscript. F. Hevia prepared the Figures. L.F. Sanz and J.A. González wrote the final version of the manuscript, reviewed by all authors.
Corresponding author
Ethics declarations
Conflict of interest
Authors declare no competing financial interest, or of any type.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Sanz, L.F., González, J.A., Hevia, F. et al. Solid–Liquid Equilibria for the Binary Systems Naphthalene or Biphenyl + 1-Tetradecanol or + 1-Hexadecanol. J Solution Chem 53, 160–181 (2024). https://doi.org/10.1007/s10953-023-01310-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10953-023-01310-2