Abstract
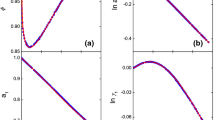
Precise vapor pressure data for solutions of NaCl, NaBr, NaI, NaClO4, KBr, KI, RbI, and CsI in methanol at 25°C in the concentration range 0.02≤m (mol-kg−1)≤0.7 are communicated and discussed. Measurements were carried out by procedures and equipment known to produce data of high precision. Polynomials of fourth degree in molalities are given for the vapor pressure of methanol solutions which can be used for calculating precise reference values as needed in indirect methods. Osmotic coefficients were calculated by taking into account the second virial coefficient of methanol vapor. Discussion of the data is based on the chemical model of electrolyte solutions taking into account short range interactions. Ion-pair association constants are compared to those of conductance measurements.
Similar content being viewed by others
References
J. Barthel, H. J. Gores, G. Schmeer, and R. Wachter, inTopics in Current Chemistry, Vol. 111, F. L. Boschke, ed. (Springer, Heidelberg, 1983).
P. Walden, H. Uhlich, and F. Laun,Z. Phys. Chem. 114, 275 (1925).
O. Kratky, H. Leopold, and H. Stabinger,Z. Angew. Phys. 27, 273 (1969).
J. Barthel and R. Neueder,GIT Fachz. Lab. 28, 1002 (1984).
J. P. Butler, H. I. Schiff, and A. R. Gordon,J. Chem. Phys. 19, 752 (1951).
A. J. Pasztor and C. M. Criss,J. Solution Chem. 7, 27 (1978).
G. Schwitzgebel, C. Luehrs, and J. Barthel,Ber. Bunsenges. Phys. Chem. 84, 1220 (1980).
J. Barthel,et al., unpublished results.
P. Beronius, J. Wikander, and A. M. Nilsson,Z. Phys. Chem. N.F. 70, 52 (1970).
A. P. Kudchadker, and P. T. Eubank,J. Chem. Eng. Data 15, 7 (1970).
H. F. Gibbard and J. L. Creek,J. Chem. Eng. Data 19, 308 (1974).
W. C. Vosburgh, L. C. Connell, and J. A. V. Butles,J. Chem. Soc., 933 (1933).
F. Kawaizumi and R. Zana,J. Phys. Chem. 78, 627 (1974).
J. Barthel, R. Neueder, F. Feuerlein, F. Strasser, and L. Iberl,J. Solution Chem. 12, 449 (1983).
J. Barthel and G. Lauermann, to be published.
R. Wachter and K. Riederer,Pure Appl. Chem. 53, 1301 (1981).
J. Barthel, inPhase Equilibria and Fluid Properties in the Chemical Industry. Part II., DECHEMA (ed.) EFCE Publ. Ser. 497 (1980).
J. Wölbl, Ph.D. Thesis, University of Regensburg (1982).
J. Barthel, R. Buchner, and H. J. Wittmann,Z. Phys. Chem. NF 139, 23 (1984).
E. C. Evers and A. G. Knox,J. Am. Chem. Soc. 73, 1739 (1951).
D. Jannakoudakis, G. Ritzoulis, and J. Roubis,Sci. Ann. Fac. Phys. Math. Aristotelian Univ. Thessaloniki 16, 255 (1976).
R. E. Jervis, D. R. Muir, J. P. Butler, and A. R. Gordon,J. Am. Chem. Soc. 75, 2855 (1953).
E. D. Copley, D. M. Murray-Rust, and H. Hartley,J. Chem. Soc. 2492 (1930).
J. E. Frazer and H. Hartley,Proc. Roy. Soc. (London) A 109, 351 (1925).
P. A. Skabichevskii,Russ. J. Phys. Chem. 43, 1432 (1969).
Y. G. Vlasov and P. P. Antonov,Russ. J. Phys. Chem. 47, 1278 (1973).
E. Bixon, R. Guerry, and D. Tassios,J. Chem. Eng. Data 24, 9 (1979).
O. F. Tower and A. F. O. Germann,J. Am. Chem. Soc. 36, 2449 (1914).
J. L. Creek, Ph.D. Thesis, University of Illinois (1975).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Barthel, J., Neueder, R. & Lauermann, G. Vapor pressures of non-aqueous electrolyte solutions. Part 1. Alkali metal salts in methanol. J Solution Chem 14, 621–633 (1985). https://doi.org/10.1007/BF00646055
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00646055