Summary
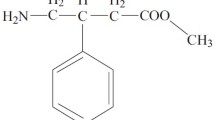
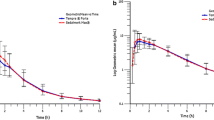
In order to evaluate the bioavailability of four different brands of phenytoin (diphenylhydantoin, DPH) tablets single doses of 600 mg DPH in acid form were given to six volunteers in a cross-over study. A micronized DPH-acid suspension was used as the reference standard. Significant differences between various products were found. The areas under the serum DPH concentration-time curves (AUC) were 26, 59, 68 and 90 per cent of the AUC of the DPH suspension. The peak serum DPH concentrations using the different tablets were 24, 54, 55 and 80 per cent of the peak value of the DPH suspension. It is likely that the differences in bio-availability of the DPH tablets are of clinical significance.
Similar content being viewed by others
References
Arnold, K., Gerber, N., Levy, G.: Absorption and dissolution studies on sodium diphenylhydantoin capsules. Canad. J. pharm. Sci.5, 89–92 (1970)
Berlin, A., Agurell, S., Borgå, O., Lund, L., Sjöqvist, F.: A micromethod for the determination of diphenylhydantoin in plasma and cerebrospinal fluid — a comparison between a gas chromatographic and a spectrophotometric method. Scand. J. clin. Lab. Invest.29, 281–287 (1972)
Duncan, D.B.: Multiple range and multiple F tests. Biometrics11, 1–42 (1955)
Finholt, P., Henriksen, G.F., Jansholt, L., Johannessen, S., Munthe-Kaas, A.W., Paulssen, R.B.: Absorption and in vitro dissolution of phenytoin and its sodium salt. Medd. norsk farm. Selsk.34, 136–150 (1972)
Glazko, A.J., Chang, T.: Diphenylhydantoin: Absorption, distribution and excretion. In: Antiepileptic drugs (eds. Woodbury, D.M., Penry, J., Schmidt, R.P.) pp. 127–136. New York: Raven Press 1972
Hall, D.G.: Hearings before the Subcommittee on Monopoly. Select Committee on Small Business, part 1 May 17, pp. 258–281. U.S. Senate, Washington, D.C.: Govt. Printing Office 1967
Johansen, H.E., Wiese, C.F.: The influence of particle size and other technological factors on the absorption and in-vitro dissolution rate of phenytoin from tablets. Arch. Pharm. Chemi77, 243–250 (1970)
Koch-Weser, J.: Bioavailability of drugs. New Engl. J. Med.291, 233–237 (1974)
Levy, G.: In: Prescription Pharmacy (ed. J.B. Sprowls), pp. 31–94. Philadelphia: J.B. Lippincott Co. 1963
Lund, L.: Clinical significance of generic inequivalence of three different pharmaceutical preparations of phenytoin. Europ. J. clin. Pharmacol.7, 119–124 (1974)
Lund, L., Alvan, G., Berlin, A., Alexanderson, B.: Pharmacokinetics of single and multiple doses of phenytoin in man. Europ. J. Clin. Pharmacol.7, 81–86 (1974)
Martin, C.M., Rubin, M., O'Malley, W.E., Garagusi, V.F., McCauley, C.E.: Comparative physiological availability of “brand” and “generic” drugs in man: chloramphenicol, sulfisoxazole, and diphenylhydantoin. Pharmacologist10, 167 (1968)
Rail, L.: “Dilantin” overdosage. Med. J. Aust.2, 339 (1968)
Tyrer, J.H., Eadie, M.J., Sutherland, J.M., Hooper, W.D.: Outbreak of anticonvulsant intoxication in an Australian city. Brit. med. J.1970 IV, 271–273
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Pentikäinen, P.J., Neuvonen, P.J. & Elfving, S.M. Bioavailability of four brands of phenytoin tablets. Eur J Clin Pharmacol 9, 213–218 (1975). https://doi.org/10.1007/BF00614020
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00614020