Summary
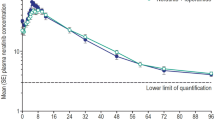
The pharmacokinetics of chlorambucil has been investigated in a cross over study after oral administration of the free drug (10 mg) and its prednisolone ester (prednimustine, 100 mg). The bioavailability of chlorambucil was about five times lower when given as prednimustine as compared to administration of the free drug. The peak plasma concentration was about twice as high and it was obtained more rapidly when the free drug was given. No intact prednimustine could be detected in plasma.
Similar content being viewed by others
References
Könyves I, Fex H, Högberg B (1973) Novel corticosteroid esters with alkylating properties. Antineoplastic Chemotherapy, Daikos GK (ed) Proc. 8th Int. Congress of Chemotherapy, 791–795
Pedersen-Bjergaard J, Mørk Hansen M, Geisler CH, Nissen NI (1980) Clinical trial of Prednimustine, Leo-1031 (NSC-134087), in patients with non-Hodgkin lymphomata and chronic lymphocytic leukaemia previously treated with steroids and alkylating agents. Acta Med Scand 207: 215–220
Johnsson JE, Tropé C, Mattsson W, Grundsell H, Aspegren K, Könyves I (1979) Phase II study of Leo 1031 (Prednimustine) in advanced ovarian carcinoma. Cancer Treat Rep 63: 421–424
Mouridsen HT, Kristensen D, Halskov Nielsen J, Dombernowsky P (1980) Phase II trial of prednimustine, L-1031, (NSC-134087) in advanced breast cancer. Cancer 46: 253–255
Kirdani RY, Murphy GP, Sandberg AA (1978) Some metabolic aspects of a nitrogen mustard of prednisolone. Oncology 35: 47–53
Plym Forshell G, Mattsson W (1981) Plasma concentrations of parent drug and metabolites after oral administration of prednimustine to patients with malignant disease. Internal Research Report, AB Leo, UP J Leo 1031 Nr 2797
Könyves I, Nordenskjöld B, Plym Forshell G, de Schryver A, Westerberg-Larsson H (1975) Preliminary clinical and absorption studies with prednimustine in patients with mammary carcinoma. Eur J Cancer 11: 841–844
Ehrsson H, Eksborg S, Wallin I, Mårde Y, Joansson B (1980) Determination of chlorambucil in plasma by GLC with selected-ion monitoring. J Pharm Sci 69: 710–712
Jakhammer T, Olsson A, Svensson L (1977) Mass fragmentographic determination of prednimustine and chlorambucil in plasma. Acta Pharm Suec 14: 485–496
Ehrsson H, Nilsson SO, Ehrnebo M, Wallin I, Wennersten G (1979) Effect of food on kinetics of 8-methoxsalen. Clin Pharmacol Ther 25 [2]: 167–171
Metzler CM (1969) NONLIN — a computer program for parameter estimation in nonlinear situations. The Upjohn Co., Technical Report No 7292/69/7292/005
Lund B, Kampmann JP, Lindahl F, Mølholm Hansen J (1976) Pivampicillin and ampicillin in bile, portal and peripheral blood. Clin Pharmacol Ther 19: 587–591
Pütter J (1979) Extramicrosomal drug metabolism. Eur J Drug Metab Pharmacokin et 4: 1–7
Gunnarsson PO, Svensson L, Wennberg B (1980) Metabolism of prednimustine by plasma enzymes. Internal Research Report, AB Leo UPJ Leo 1031 Nr 2583
Wilkinson R. Gunnarsson PO, Plym Forshell G, Renshaw J, Harrap KR (1978) The hydrolysis of prednimustine by enzymes from normal and tumour tissues. Characterization and treatment of human tumours. Maltoni C (ed). Excerpta Medica International Congress Series 420: 260–273
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Ehrsson, H., Wallin, I., Nilsson, S.O. et al. Pharmacokinetics of chlorambucil in man after administration of the free drug and its prednisolone ester (prednimustine, Leo 1031). Eur J Clin Pharmacol 24, 251–253 (1983). https://doi.org/10.1007/BF00613827
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00613827