Summary
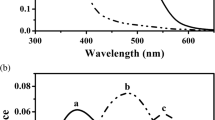
The visual pigments of four mesopelagic crustacean species were studied at sea by means of microspectrophotometry. The absorbance maxima obtained for the visual pigments and their metarhodopsins, respectively, were: 493 nm and 481 nm (Systellaspis debilis), 485 nm and 480 nm (Acanthephyra curtirostris), 491 nm and 482 nm (A. smithi), and 495 nm and 487 nm (Sergestes tenuiremis). The spectral characteristics of the rhodopsins and metarhodopsins permit high photosensitivity and facilitate photoregeneration in a nearly monochromatic environment. Photic regeneration of rhodopsins from the deep-sea environment was demonstrated, and data were obtained which are consistent with the occurrence of dark regeneration. Specific optical density of the observed visual pigments was calculated for two species.
Similar content being viewed by others
References
Bernard GD (1983) Dark-processes following photoconversion of butterfly rhodopsins. Biophys Struct Mech 9:277–286
Bruno MS (1971) Microspectrophotometry of isolated crustacean rhabdoms. PhD thesis, Harvard University
Bruno MS, Goldsmith TH (1974) Rhodopsin of the blue crabCallinectes: evidence for absorption differences in vitro and in vivo. Vision Res 14:653–658
Bruno MS, Mote MI, Goldsmith TH (1973) Spectral absorption and sensitivity measurements in single ommatidia of the green crab,Carcinus. J Comp Physiol 82:151–163
Bruno MS, Barnes SN, Goldsmith TH (1977) The visual pigment and visual cycle of the lobster,Homarus. J Comp Physiol 120:123–142
Childress JJ, Barnes AT, Quetin LB, Robison BH (1978) Thermally protecting cod ends for the recovery of living deep-sea animals. Deep-Sea Res 25:419–422
Crescitelli F, McFall-Ngai M, Horwitz J (1985) The visual pigment sensitivity hypothesis: further evidence from fishes of varying habitats. J Comp Physiol A 157:323–333
Cronin TW, Goldsmith TH (1982) Quantum efficiency and photosensitivity of the rhodopsin⇌metarhodopsin conversion in crayfish photoreceptors. Photochem Photobiol 36:447–454
Cronin TW, Goldsmith TH (1984) Dark regeneration of rhodopsin in crayfish photoreceptors. J Gen Physiol 84:63–81
Cummins D, Goldsmith TH (1981) Cellular identification of the violet receptor in the crayfish eye. J Comp Physiol 142:199–202
Dartnall HJA (1953) The interpretation of spectral sensitivity curves. Br Med Bull 9:24–30
Dartnall HJA (1975) Assessing the fitness of visual pigments for their photic environments. In: Ali MA (ed) Vision in fishes. Plenum Press, New York London, pp 543–563
Dartnall HJA, Lythgoe JN (1965) The spectral clustering of visual pigments. Vision Res 5:81–100
Denton EJ, Shaw TI (1963) The visual pigments of some deepsea elasmobranchs. J Mar Biol Ass UK 43:65–70
Denton EJ, Warren FJ (1956) Visual pigments of deep-sea fish. Nature (Lond) 178:1059
Denton EJ, Warren FJ (1957) The photosensitive pigments in the retinae of deep-sea fish. J Mar Biol Ass UK 36:651–662
Denys CJ, Brown PK (1982) The rhodopsins ofEuphausia superba andMeganyctiphanes norvegica (Crustacea, Euphausiacea). J Gen Physiol 80:451–472
Fernandez HR (1978) Visual pigments of bioluminescent and nonbioluminescent deep-sea fishes. Vision Res 19:589–592
Fisher LR, Goldie EH (1960) Pigments of compound eyes. In: Christensen B, Buchmann B (eds) Progress in photobiology. Elsevier, Amsterdam London New York Princeton, pp 153–154
Frank TM (1986) Visual spectral sensitivity of deep-sea decapod crustaceans. Am Zool 26:35A
Goldsmith TH (1972) The natural history of invertebrate visual pigments. In: Dartnall HJA (ed) Photochemistry of vision (Handbook of sensory physiology, vol. VII/1). Springer, Berlin Heidelberg New York, pp 685–719
Goldsmith TH (1978a) The effects of screening pigments on the spectral sensitivity of some Crustacea with scotopic (superposition) eyes. Vision Res 18:475–482
Goldsmith TH (1978b) The spectral absorption of crayfish rhabdoms: pigment, photoproduct, and pH sensitivity. Vision Res 18:463–473
Goldsmith TH, Wehner R (1977) Restrictions on rotational and translational diffusion of pigment in the membranes of a rhabdomeric photoreceptor. J Gen Physiol 70:453–490
Hamacher K, Stieve H (1984) Spectral properties of the rhodopsin-system of the crayfishAstacus leptodactylus. Photochem Photobiol 39:379–390
Herring PH (1978) Bioluminescence in action. Acadmic Press, London New York San Francisco
Herring PJ (1983) The spectral characteristics of luminous marine organisms. Proc R Soc Lond B 220:183–217
Jerlov NG (1976) Marine optics. Elsevier Oceanography Series, vol 14. Elsevier Scientific, Amsterdam Oxford New York, 231 pp
Kampa EM (1955) Euphausiopsin, a new photosensitive pigment from the eyes of euphausiid crustaceans. Nature (Lond) 175:996–998
Land MF (1976) Superposition images are formed by reflection in the eyes of some oceanic decapod Crustacea. Nature 263:764–765
Land MF (1981) Optics and vision in invertebrates. In: Autrum H (ed) Vision in invertebrates (Handbook of sensory physiology, vol VII/6B). Springer, Berlin Heidelberg New York, pp 471–592
Land MF, Burton FA, Meyer-Rochow VB (1979) The optical geometry of euphausiid eyes. J Comp Physiol 130:49–62
Loew ER (1976) Light and photoreceptor degeneration in the Norway lobsterNephrops norvegicus (L.). Proc. R Soc Lond B 193:31–44
Lythgoe JN (1972) List of vertebrate visual pigments. In: Dartnall HJA (ed) Photochemistry of vision (Handbook of sensory physiology, vol VII/2). Springer, Berlin Heidelberg New York, pp 604–624
Munz FW (1958) Photosensitive pigments from the retinae of certain deep-sea fishes. J Physiol 140:220–235
Nilsson HL (1982) Rhabdom breakdown in the eye ofCirolana borealis (Crustacea) caused by exposure to daylight. Cell Tissue Res 227:633–639
Nilsson HL, Lindstrom M (1983) Retinal damage and sensitivity loss of a light-sensitive crustacean compound eye (Ciroana borealis): electron microscopy and electrophysiology. J Exp Biol 107:277–292
O'Day WT, Fernandez HR (1974)Aristostomias scintellans (Malacosteidae): a deep-sea fish with visual pigments apparently adapted to its own bioluminescence. Vision Res 14:545–550
O'Day WT, Fernandez HR (1976) Vision in the lanternfishStenobrachius leucopsarus (Myctophidae). Mar Biol 37:187–195
Savitzky A, Golay MJE (1964) Smoothing and differentiation of data by simplified least squares procedures. Analyt Chem 36:1627–1639
Schwemer J (1983) Pathways of visual pigment regeneration in fly photoreceptor cells. Biophys Struct Mech 9:287–298
Schwemer J (1984) Renewal of visual pigment in photoreceptors of the blowfly. J Comp Physiol A 154:535–547
Shelton PMJ, Gaten E, Chapman CJ (1985) Light and retinal damage inNephrops norvegicus (L.) (Crustacea). Proc R Soc Lond B 226:217–236
Stavenga DG, Schwemer J (1984) Visual pigments of invertebrates. In: Ali MA (ed) Photoreception and vision in invertebrates. Plenum Press, New York, pp 11–62
Stowe S (1980) Spectral sensitivity of retinal pigment movement in the crabLeptograpsus variegatus (Fabricius). J Exp Biol 87:73–98
Vogt K, Kirschfeld K, Stavenga DG (1982) Spectral effects of the pupil in fly receptors. J Comp Physiol 146:145–152
Wald GW, Brown PK, Brown PS (1957) Visual pigments and depths of habitat of marine fishes. Nature (Lond) 180:969–971
Walters JF (1975) Ecology of Hawaiian sergestid shrimps (Penaeidea: Sergestidae). PhD thesis, University of Hawaii, 227 pp
Widder EA, Latz MI, Case JF (1983) Marine bioluminescence spectra measured with an optical multichannel detection system. Biol Bull 165:791–810
Widder EA, Hiller-Adams P, Case JF (1987) A multichannel microspectrophotometer for visual pigment investigations. Vision Res 27:1047–1055
Yaldwyn JC (1957) Deep-water Crustacea of the genusSergestes (Decapoda, Natantia) from Cook Strait, New Zealand. Zoological Publications from Victoria University of Wellington 22:1–27
Ziemann DA (1975) Patterns of vertical distribution, vertical migration, and reproduction in the Hawaiian mesopelagic shrimps of the family Oplophoridae. PhD thesis, University of Hawaii
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Hiller-Adams, P., Widder, E.A. & Case, J.F. The visual pigments of four deep-sea crustacean species. J. Comp. Physiol. 163, 63–72 (1988). https://doi.org/10.1007/BF00611997
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00611997