Abstract
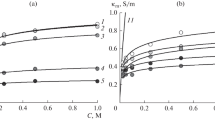
By using cation-exchange membranes prepared by the paste method, a study was made of the influence of the distribution of polyvinyl chloride (PVC) on the electrical conductivity of the membranes and on the structure of the cation-exchange resin component (R-resin). Four kinds of cation-exchange membranes with different PVC contents and degrees of cross-linking were used. The experimental method involved treatment with hydrogen peroxide for selective decomposition of the R-resin. The estimated ratio of the specific electrical conductivity of the R-resin to that of the membrane varied from 2.2 to 4.4, depending upon the volume ratio of PVC in the membrane (0.33–0.45). Each of the membranes had a very high concentration of ion-exchange groups for cross-linking. This could be explained in terms of the mechanical and chemical restraint imposed by the PVC against the swelling of the R-resin. The structure of the R-resin, however, was almost independent of the PVC volume ratio over the range investigated.
Similar content being viewed by others
References
Y. Mizutani,Bull. Chem. Soc. Japan 43 (1970) 595.
Y. Mizutani and M. Nishimura,J. Appl. Polymer Sci. 14 (1970) 1847.
Y. Mizutani, W. Teshima and S. Akiyama,Japanese Patent 410 368 (1963).
Y. Mizutani R. Yamane and H. Motomura,Japanese Patent 448 778 (1965).
Y. Mizutani W. Teshima, S. Akiyama, R. Yamane and H. Ihara,US Patent 3451 951 (1969).
H. Shimizu and Y. Kosaka, ‘Ionkōkan-Maku (Ion Exchange Membranes)’, Kyōritsu Publishing Co., Tokyo (1963) p. 117.
Y. Onoue, Y. Mizutani and R. Yamane,Denki-Kagaku (J. Electrochem. Soc. Japan) 27 (1959) 482.
H. Kawabe, H. Jacobson, I. F. Miller and H. P. Gregor,J. Colloid Interface Sci. 21 (1966) 79.
K. S. Spiegier,J. Electrochem. Soc. 113 (1966) 161.
J. E. Salmon,Disc. Faraday Soc. 21 (1956) 123.
H. P. Gregor, in ‘Saline Water Conversion’ (ited by Mason) Publ 568, National Academy of Sciences, National Research Council, Washington, DC (1958) p. 242.
D. K. Hale and D. J. McCauley,Trans. Faraday Soc. 57 (1961) 135.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Nishimura, M., Mizutani, Y. Correlation between structure and properties of cation-exchange membranes prepared by the paste method. J Appl Electrochem 11, 165–171 (1981). https://doi.org/10.1007/BF00610976
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00610976