Abstract
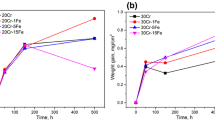
Additions of 3% silicon or manganese to Ni-20 Cr reduced the oxidation rate, whereas additions of 1% had little effect. Three percent silicon alloys formed an inner scale of SiO2, and 3% manganese alloys formed an inner spinel layer of essentially pure MnCr2O4. The experimentally determined solid-state growth rate of NiCr2O4 was about 1000 times slower than the growth rate for Cr2O3. It has been established that the protective layer on Ni-20 Cr (Nichrome alloys) is the spinel and not Cr2O3 as previously postulated. The mechanism for scale growth is discussed for Ni-20 Cr alloys.
Similar content being viewed by others
References
J. W. Hickman and E. A. Gulberansen,Trans. AIME 180, 519 (1949).
M. Sugiyama and T. Nakayama,Japan Inst. Metals J. 24, 541 (1960).
E. A. Gulbransen and W. R. McMillan,Anal. Eng. Chem. 45, 1734 (1953).
E. A. Gulbransen and K. F. Andrew,J. Electrochem. Soc. 106, 941 (1959).
G. C. Wood and T. Hodgkiess,J. Electrochem. Soc. 113, 319 (1966).
G. C. Wood, T. Hodgkiess, and D. P. Whittle,Corrosion Sci. 6, 129 (1966).
N. Birks and H. Rickert,J. Inst. Metals 91, 308 (1962–63).
J. S. Armijo, Report NASA-CR72537 (1969).
P. Kofstad,High Temperature Oxidation of Metals (Wiley, New York, 1966), p. 166.
C. Wagner,Z. Phys. Chem. 34, 309 (1936).
H. Schmalzried.Z. Phys. Chem. (N.F.)33, 111 (1962).
H. Schmalzried,Ber. Deut. Keram. Ges. 42, 11 (1965).
E. B. Evans, C. A. Phalnikar, and W. M. Baldwin, Jr.,J. Electrochem. Soc. 103, 367 (1956).
I. S. Gil'dengorn and I. L. Rogel'berg,Fiz. Metal, i Metalloved. 18, 935 (1964).
Author information
Authors and Affiliations
Additional information
This work was performed at Stanford Research Institute, Menlo Park, Calif. and was supported by the National Aeronautics amd Space Administration, Contract NAS 3-11165.
Rights and permissions
About this article
Cite this article
Douglass, D.L., Armijo, J.S. The effect of silicon and manganese on the oxidation mechanism of Ni-20 Cr. Oxid Met 2, 207–231 (1970). https://doi.org/10.1007/BF00603657
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00603657