Abstract
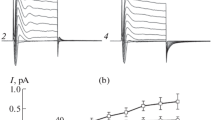
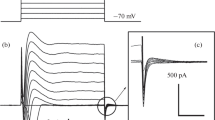
The intracellular chloride activity and its dependence on ionic substitutions in the bathing media was studied in individual surface cells of resting gastric mucosa using conventional and Cl− selective microelectrodes. When the tissue was perfused with control NaCl-Ringer the cell membrane p.d.'s, cell-lumen (Ψcm) and cell-serosa (Ψcs) were −40.9±0.6 mV and −66.8±0.5 mV (n=175) respectively specitively and the p.d. measured by the Cl− selective microelectrodes across the serosal membrane (Ψ Cl-cs ) averaged −32.4±0.7 mV (n=138). From these values an intracellular Cl− activity (a Cl−c ) of 15.3 mmol/l can be estimated. The data indicate that chloride ion is distributed close to equilibrium at the luminal membrane while it is accumulated by an energy requiring step at the serosal membrane. Reduction (2 mmol/l) or absence of chloride from the luminal bath did not result in any detectable change ofa Cl−c ; on the other hand, after removal of Cl− from the serosal bath the intracellular Cl− activity fell to 7.1 mmol/l.
When the tissue was exposed to serosal Na+-free Ringer (Na+ replaced by choline or TMA), although thea Cl−c remained unaffected, a marked reduction of the electrochemical gradient for Cl− at the serosal membrane was observed.
These data indicate that: (i) chloride is accumulated in the surface cells against its electrochemical potential difference at the serosal membrane; (ii) the luminal membrane has a negligible conductance to Cl−, while the serosal membrane represents a conductive pathway to chloride; (iii) the uphill entry of chloride at the serosal membrane seems to be, at least partially, Na+-dependent.
Similar content being viewed by others
References
Armstrong WMcD, Garcia-Diaz JF, O'Doherty J, O'Regan MG (1979) Transmucosal Na+ electrochemical potential difference and solute accumulation in epithelial cells of the small intestine. Fed Proc 38:2722–2728
Blum AL, Hirschowitz BI, Helander HF, Sachs G (1971) Electrical properties of isolated cells of Necturus gastric mucosa. Biochim Biophys Acta 241:261–272
Carrasquer G, Chu TC, Rehm WS, Schwartz M (1982) Evidence for electrogenic Na−Cl symport in the in vitro frog stomach. Am J Physiol 262:G620-G627
Cassola AC, Mollenhauer M, Frömter E (1983) The intracellular chloride activity of rat kidney. Pflügers Arch 399:259–265
Duffey ME, Turnheim K, Frizzell RA, Schultz SG (1978) Intracellular chloride activity in rabbit gallbladder: direct evidence for the role of the sodium gradient in energizing “uphill” chloride transport. J Membr Biol 42:229–245
Duffey ME, Thompson SM, Frizzell RA, Schultz SG (1979) Intracellular chloride activities and active chloride absorption in the intestinal epithelium of the winter flounder. J Membr Biol 50:331–341
Edelman A, Curci S, Samarizija I, Frömter E (1978) Determination of intracellular K+ activity in rat kidney proximal tubular cells. Pflügers Arch 378:37–45
Flemström G (1977) Active alkalinization by amphibian gastric fundic mucosa in vitro. Am J Physiol 233:E1-E12
Forte JG, Limlomwongse L, Kasbekar DK (1969) Ion transport and the development of hydrogen ion secretion in the stomach of the metamorphosing bullfrog tadpole. J Gen Physiol 54:79–95
Forte JG, Machen TE (1975) Transport and electrical phenomena in resting and secreting piglet gastric mucosa. J Physiol (Lond) 244:33–51
Frizzell RA, Field M, Schultz SG (1979) Sodium-coupled chloride transport by epithelial tissues. Am J Physiol 236:F1-F8
Frizzell RA, Smith PL, Vosburgh E, Field M (1979) Coupled sodium-chloride influx across brush border of flounder intestine. J Membr Biol 46:27–39
Greger R (1981) Coupled transport of Na+ and Cl− in the thick ascending limb of Henle's loop of rabbit nephron. Scand Audiol (Suppl) 14:1–15
Greger R, Schlatter E, Lang F (1983) Evidence for electroneutral sodium chloride cotransport in the cortical thick ascending limb of Henle's loop of rabbit kidney. Pflügers Arch 396:308–314
Helander HF, Sanders SS, Rehm WS, Hirschowitz BI (1972) Quantitative aspects of gastric morphology. In: Sachs G, Heinz E, Ullrich KJ (eds) Gastric secretion. Academic Press, New York, pp 69–88
Hodgkin AL, Horowicz P (1959) The influence of potassium and chloride ions on the membrane potential of single muscle fibers. J Physiol (Lond) 148:127–160
Hogben CAM (1955) Active transport of chloride by isolated frog gastric epithelium. Origin of the gastric mucosal potential. Am J Physiol 180:641–649
Ito S (1967) Anatomic structure of the gastric mucosa. In: Handbook of physiology. Alimentary canal, vol II. Am Physiol Soc, Washington DC, pp 705–741
Liedtke CM, Hopfer U (1982) Mechanism of Cl− translocation across small intestinal brush border membrane. II. Demonstration of Cl−−OH− exchange and Cl− conductance. Am J Physiol 242:G272-G280
Machen TE, Mc Lennan WL (1980) Na+-dependent H+ and Cl− transport in in vitro frog gastric mucosa. Am J Physiol 238:G403-G413
Machen TE, Zeuthen T (1982) Cl− transport by gastric mucosa: cellular Cl− activity and membrane permeability. Phil Trans R Soc L 299:559–573
Norris JL (1959) The normal histology of the esophageal and gastric mucosal of the frog,Rana pipiens. J Exp Zool 141:155–161
Norris SH, Jackson MJ (1982) Electrogenic and non electrogenic transport across rat gastric mucosa. (Abstract.) Fed Proc 41:4158
Sachs G, Spenney JG, Lewin M (1978) H+-Transport: regulation and mechanism in gastric mucosa and membrane vesicles. Physiol Rev 58:106–173
Schettino T, Cremaschi D, Lippe C, Lora Lamia Donin C, Cotelli F (1980) Non-electrolyte fluxes across gastric mucosa in relation to gastric stimulation. Is gastric juice secreted by osmosis or exocytosis? Pflügers Arch 387:269–279
Schettino T, Curci S (1980) Intracellular potassium activity in epithelial cells of frog fundic gastric mucosa. Pflügers Arch 383:99–103
Shoemaker RL, Sachs G (1972) Microelectrode studies of Necturus gastric mucosa. In: Sachs G, Heinz E, Ullrich KJ (eds) Gastric secretion. Academic Press, New York, pp 147–157
Silva P, Stoff J, Field M, Fine L, Forrest JN, Epstein FH (1977) Mechanism of active chloride secretion by shark rectal gland: role of Na−K ATPase in chloride transport. Am J Physiol 233:F298-F306
Spring KR, Kimura G (1978) Chloride reabsorption by renal proximal tubules of Necturus. J Membr Biol 38:233–254
Vulliemin P, Durand-Archzynska W, Durand J (1983) Electrical properties and electrolyte transport in bovine tracheal epithelium: effects of ion substitutions, transport inhibitors and histamine. Pflügers Arch 396:54–59
Welsh MJ, Smith PL, Frizzell RA (1982) Chloride secretion by canine tracheal epithelium. II. The cellular electrical potential profile. J Membr Biol 70:227–238
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Curci, S., Schettino, T. Effect of external sodium on intracellular chloride activity in the surface cells of frog gastric mucosa. Pflugers Arch. 401, 152–159 (1984). https://doi.org/10.1007/BF00583875
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00583875