Abstract
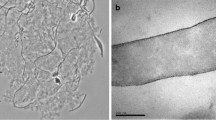
Enrichments on L-tartrate from a freshwater lake sediment yielded a pure culture of anaerobic bacterium designated strain 16Lt1. The rod-shaped organism was motile, did not form spores, and had a gram-negative wall structure. No cytochromes were detected. The mol % G+C of the DNA was 58. The new strain was microaerotolerant, and grew optimally at 30°C and neutral pH in freshwater medium. A wide range of carbohydrates was fermented, with formate, acetate, ethanol, lactate and succinate being the end-products detected. L-tartrate and citrate were fermented to formate, acetate and CO2. L-tartrate was fermented by the dehydratase pathway, and glucose by the Embden-Meyerhof-Parnas pathway. Fumarate was reduced, but nitrate, sulfate, sulfur and thiosulfate were not used as terminal electron acceptors. Glucose metabolism was constitutive, whereas L-tartrate-degrading activity was inducible. When glucose and L-tartrate were both present as substrates, growth was diauxic with glucose being metabolized first. The growth rate and growth yield were higher on glucose than on L-tartrate. Strain 16Lt1 has been deposited with the Deutsche Sammlung von Mikroorganismen as ‘Bacteroides’ sp. DSM6268.
Similar content being viewed by others
References
Anthony C & Guest JR (1968) Deferred metabolism of glucose byClostridium tetanomorphum. J. Gen. Microbiol. 54: 277–286
Blenden DC & Goldberg HS (1965) Silver inpregnation stain forLeptospira and flagella. J. Bacteriol. 89: 899–900
Cerny G (1978) Studies on the aminopeptidase test for the distinction of Gram-negative from Gram-positive bacteria. Eur. J. Appl. Microbiol. Biotechnol. 5: 113–122
Cogan TM (1987) Co-metabolism of citrate and glucose byLeuconostoc spp.: Effects on growth, substrates and products. J. Appl. Bacteriol. 63: 551–558
deVries W, Rietveld-Struijk TRM & Stouthamer AH (1977) ATP formation associated with fumarate and nitrate reduction in growing cultures ofVeillonella alcalescens. Antonie van Leeuwenhoek 43: 153–167
Doelle HW (1975) Bacterial Metabolism, 2nd edition. Academic Press, New York
Holdeman LV, Kelley RW, Moore WEC (1984)Bacteroides. In: Krieg NR, Holt JG (Eds) Bergey's Manual of Systematic Bacteriology, Vol 1 (pp 604–631). Williams and Wilkins, Baltimore
Janssen PH & Harfoot CG (1990a). Isolation of aCitrobacter species able to grow on malonate under strictly anaerobic conditions. J. Gen. Microbiol. 136: 1037–1042
(1990b)Ilyobacter delafieldii sp. nov., a metabolically restricted anaerobic bacterium fementing PHB. Arch. Microbiol. 154: 253–259
Kandler O & Weiss N (1986)Lactobacillus. In: Sneath PHA, Mair NS, Sharpe ME, Holt JG (Eds) Bergey's Manual of Systematic Bacteriology, Vol 2 (pp 1209–1234). Williams and Wilkins, Baltimore, Maryland
Kohn LD, Hurlbert RE & Jakoby WB (1974) L(+)-tartrate and meso-tartrate. In: Bergmeyer HU (Ed) Methods of Enzymatic Analysis, Vol 3, 2nd English edition (pp 1397–1403). Verlag Chemie, Weinheim
Kornberg HL & Reeves RE (1972) Inducible phosphoenolpyruvate-dependent hexose phosphotransferase activities inEscherichia coli. Biochem. J. 128: 1339–1344
Lamed R & Zeikus JG (1980) Glucose fermentation pathway ofThermoanaerobium brockii. J. Bacteriol. 141: 1251–1257
Lee IH, Frederickson AG & Tsuchiya HM (1974) Diauxic growth ofPropionibacterium shermanii. Appl. Microbiol. 28: 831–835
Mavrovouniotis ML, Bayol P, Lam TKM, Stephanopoulos G & Stephanopoulos G (1988) A group contribution method for the estimation of equilibrium constants for biochemical reactions. Biotechnol. Tech. 2: 23–28
Norris JR, Berkeley RCW, Logan NA & O'Donnell AG (1981) The generaBacillus andSporolactobacillus. In: Starr MP, Stolp H, Trüper HG, Balows A, Schlegel HG (Eds) The Prokaryotes: A Handbook on Habitats, Isolation and Identification of Bacteria, Vol 2 (pp 1711–1742). Springer-Verlag, Berlin Heidelberg New York
Radler F & Yannissis C (1972) Weinsäureabbau bei Milchsäurebakterien. Arch. Mikrobiol. 82: 219–239
Russell JB & Baldwin RL (1978) Substrate preferences in rumen bacteria: Evidence of catabolite regulatory mechanisms. Appl. Environ. Microbiol. 36: 319–329
Schink B (1984) Fermentation of tartrate enantiomers by anaerobic bacteria and description of two new species of strict anaerobes,Ruminococcus pasteurii andIlyobacter tartaricus. Arch. Microbiol. 139: 409–414
Scopes R (1982) Protein Purification: Principles and Practice. Springer-Verlag, New York Heidelberg Berlin
Shah HN & Collins MD (1989) Proposal to restrict the genusBacteroides (Castellani and Chalmers) toBacteroides fragilis and closely related species. Int. J. System. Bacteriol. 39: 85–87
Stouthamer AH (1979) The search for correlation between theoretical and experimental growth yields. In: Quayle JR (Ed) International Review of Biochemistry, Vol 21: Microbial Biochemistry (pp 1–47). University Park Press, Baltimore, Maryland
Thauer RK, Jungermann K & Decker K (1977) Energy conservation in chemotrophic anaerobic bacteria. Bacteriol. Rev. 41: 100–180
Wagener S & Pfennig N (1987) Monoxenic culture of the anaerobic ciliateTrimyema compressum Lackey. Arch. Microbiol. 149: 4–11.
Wagener S, Bardele CF & Pfennig N (1990) Functional integration ofMethanobacterium formicicum into the anaerobic ciliateTrimyema compressum. Arch. Microbiol. 153: 496–501
Windholz M, Budavari S, Stroumtsos LY & Fertig MN (1976) The Merck Index, 9th edition. Merck, Rahway, New Jersey
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Janssen, P.H. Fermentation of L-tartrate by a newly isolated gram-negative glycolytic bacterium. Antonie van Leeuwenhoek 59, 191–198 (1991). https://doi.org/10.1007/BF00580659
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00580659