Summary
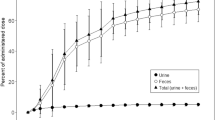
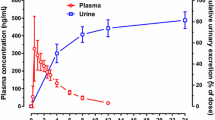
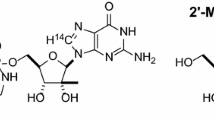
The absorption and elimination kinetics of 4-amino-N10-methylpteroyl-D-glutamic acid (D-MTX), the optical isomer of methotrexate (L-MTX), were examined. Test doses of 10 mg D-MTX were administered i.v. and orally to nine patients and its plasma concentration and urinary excretion were followed. The plasma curves after an i.v. bolus injection of D-MTX declined strictly biexponentially and reached zero after about 16 h. The elimination rate constants were estimated as the terminal first order rate constants. The absorption of orally administered D-MTX, estimated by its 24 h urinary recovery, in all cases was less than 3% of the dose administered. The insignificant intestinal absorption made it possible to estimate the renal and biliary secretion rates of D-MTX from the overall elimination rate constant and from the fraction of the dose excreted in urine. In three of the patients, elimination rate constants both for D-MTX and L-MTX were obtained. The renal elimination rates of the two compounds were found to be nearly identical. The median ratio of biliary/renal excretion of D-MTX was 0.94 (range 0.41–1.50), which indicates extensive entero-hepatic cycling and active reabsorption of L-MTX at the therapeutic dose levels used in psoriasis.
Similar content being viewed by others
References
Baird GM, Dossetor JFB (1981) Methotrexate enteropathy. Lancet 1: 164
Baugh CM, Krumdieck CL, Nair MG (1973) Polyglutamyl metabolites of methotrexate. Biochem Biophys Res Commun 52: 27–34
Bedi TR, Bhutani LK, Kandhari KC, Tandon BN (1974) Small bowel in skin diseases. Ind J Med Res 62: 142–143
Bender RA, Makulu DR (1976) Relationship between intracellular dihydrofolate reductase and tightly bound intracellular methotrexate in human neoplastic cells. Biochem Pharmacol 25: 975–976
Bertino JR, Cashmore A, Fink M, Calabresi P, Lefkowitz E (1965) The induction of leucocyte and erythrocyte dihydrofolate reductase by methotrexate. Clin Pharmacol Ther 6: 763–770
Bischoff KB, Dedrick RL, Zaharko DS, Longstreth JA (1971) Methotrexate pharmacokinetics. J Pharm Sci 60: 1128–1133
Bleyer WA (1978) The clinical pharmacology of methotrexate. Cancer 41: 36–51
Calvert AH, Bondy PK, Harrap KP (1977) Some observations on the human pharmacology of methotrexate. Cancer Treat Rep 61: 1647–1656
Creaven PJ, Hansen HH, Alford DA, Allen LM (1973) Methotrexate in liver and bile after intravenous dosage in man. Br J Cancer 28: 589–591
Hendel J, Sarek LJ, Hvidberg EF (1976) Rapid radioimmunoassay for methotrexate in biological fluids. Clin Chem 22: 813–816
Hendel J, Sarek L (1977) Production of methotrexate antiserum in rabbits: the significance of immunogen solobility, hapten content, and mode of administration on the antibody response. Scand J Clin Lab Invest 37: 273–278
Hendel J (1978) Intracellular metabolites of methotrexate. Chemother Oncol 2[Suppl]: 135–140
Hendel L, Hendel J, Johansen A, Gudmand-Hoyer E (1982) Intestinal function and methotrexate absorption in psoriatic patients. Clin Exp Dermatol 7: 491–498
Henderson ES, Adamson RH, Oliverio VT (1965) The metabolic fate of tritiated methotrexate II. Absorption and excretion in man. Cancer Res 25: 1018–1024
Hoede N, Morches B, Holzmann H (1974) Psoriasis — eine All-gemeinerkrankung. Internist 15: 186–191
Lee WW, Martinez AP, Goodman L (1974) Methotrexate analogs containing spurious amino acids. J Med Chem 17: 326–330
Shuster S, Watson AJ, Marks J (1967) Small intestine in psoriasis. Br Med J 3: 458–460
Wan SH, Huffman DH, Azarnoff DL, Stephens R, Hoogstraten B (1974) Effect of route of administration and effusions on methotrexate pharmacokinetics. Cancer Res 34: 3487–3491
Whitehead VM, Perrault MM, Stelcner S (1975) Tissue-specific synthesis of methotrexate polyglutamates in the rat. Cancer Res 35: 2985–2990
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Hendel, J., Brodthagen, H. Entero-hepatic cycling of methotrexate estimated by use of the D-isomer as a reference marker. Eur J Clin Pharmacol 26, 103–107 (1984). https://doi.org/10.1007/BF00546716
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00546716