Abstract
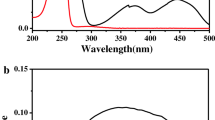
Absorption and emission spectroscopy, utilizing theoretical group analysis, was used to investigate the nature of triplet states of chrornate and bichromate ions in aqueous solutions. The complex kinetics observed for the quenching of phosphorescence is explained by competition of emission and rapid exchange of energy between triplet sublevels. Quenching of phosphorescence is observed in concentrated chromate ion solutions. The dimer nature of the bichromate ion prevents intermolecular exchange of energy between ions in view of the existence of a more effective intraionic energy transfer.
Similar content being viewed by others
References
J. A. Cannor, I. H. Hiller, V. R. Saunders, et al., Mol. Phys., 24, No. 3, 497–509 (1972).
Z. G. Szabo, K. Kamaras, Sz. Szebeni, and I. Ruft, Spectrochim. Acta, 34, No. 6, 607–612 (1978).
T. Ziegler, A. Rauk, and E. I. Baerends, Chem. Phys., 16, No. 2, 209–217 (1976).
A. I. Kryukov, V. P. Sherstyuk, and I. I. Dilung, Phototransfer of an Electron and Its Applied Aspects [in Russian], Nauk. Dumka, Kiev (1982).
N. A. Kondratenko and V. P. Sherstyuk, Teor. Éksp. Khim., 22, No. 6, 686–693 (1986).
L. M. Jonson and S. P. McGlynn, Chem. Phys. Lett., 7, No. 6, 618–620 (1970).
J. H. van der Waals, W. A. J. A. van der Poel, J. Luminescence, 31/32, Pt. 1, 123–125 (1984).
M. U. Bel'sh, Yu. D. Glinka, I. Ya. Kushnirenko, et al., Spectroscopy of Condensed Media [in Russian], Kiev (1988), pp. 92–96.
Yu. V. Plyuto, Yu. I. Gorlov, and A. A. Chuiko, Dokl. Akad. Nauk UkrSSR, Ser. B., No. 11, 53–55 (1983).
Author information
Authors and Affiliations
Additional information
Translated from Teoreticheskaya i Éksperimental'naya Khimiya, Vol. 30, No. 6, pp. 337–341, November–December, 1994.
Rights and permissions
About this article
Cite this article
Kondratenko, N.A., Piryatinskii, Y.P., Sherstyuk, V.P. et al. Nature and kinetics of the self-quenching of excited states of chromium(VI) oxyanions. Theor Exp Chem 30, 294–297 (1995). https://doi.org/10.1007/BF00538127
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00538127