Abstract
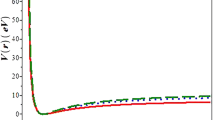
The possibility of using the virial theorem to calculate the coefficients of the potential function for diatomic hydrides on the basis of the Fock-Roothaan wave function in the closed electron shell approximation has been demonstrated in relation to the LiH, BH, CH, NH, and OH molecules.
Similar content being viewed by others
References
M. L. Clinton, J. Chem. Phys., 33, 1603, 1960.
P. Phillipson, J. Chem. Phys., 39, 3010, 1963.
L. Salem, J. Chem. Phys., 38, 1227, 1963.
M. L. Benston and B. Kirtman, J. Chem. Phys., 44, 119, 1966.
P. Phillipson, J. Chem. Phys., 44, 633, 1966.
W. R. Ross and P. Phillipson, J. Chem. Phys., 44, 844, 1966.
R. H. Schwendeman, J. Chem. Phys., 44, 556, 1966.
R. H. Schwendeman, J. Chem. Phys., 44, 2115, 1966.
C. C. I. Roothaan, Rev. Mod. Phys., 23, 69, 1951.
W. Kolos and C. C. I. Roothaan, Rev. Mod. Phys., 32, 219, 1960.
M. Cohen and A. Dalgarno, Proc. Phys. Soc., 77, 748, 1961.
W. Kauzmann, An Introduction to Quantum Chemistry [Russian translation], Moscow, IL, 1960.
V. V. Rossikhin and V. P. Morozov, TEKh [Theoretical and Experimental Chemistry], 2, 528, 1966.
Yu. A. Kruglyak and D. K. Witman, Tables of Integrals for Quantum Chemistry [in Russian], Moscow, 1965.
R. K. Nesbet, J. Chem. Phys., 40, 3619, 1964.
B. I. Ransil, Rev. Mod. Phys., 32, 245, 1960.
M. Krauss, J. Chem. Phys., 28, 1021, 1958.
G. Herzberg, Diatomic Molecules, D. Van Nostrand Company, Inc., Princeton, New Jersey, 1955.
J. Goodisman, J. Chem. Phys., 38, 304, 1963.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Rossikhin, V.V., Morozov, V.P. & Bezzub, L.I. Use of the virial theorem to calculate the molecular force constants of diatomic hydrides. Theor Exp Chem 4, 22–24 (1970). https://doi.org/10.1007/BF00525940
Issue Date:
DOI: https://doi.org/10.1007/BF00525940