Abstract
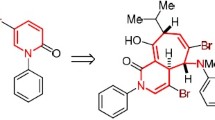
The stereochemistry of adducts of aryloxyfurans with maleic acid derivatives was studied by PMR spectroscopy. It was shown that adducts with maleic anhydride are produced only in the form of exo isomers, whereas adducts with N-phenyl-maleinimide are isolated from the reaction in the form of mixtures of endo and exo forms. Bromination of the adducts was realized. The orientation of the bromine atoms in the bromination products was established by PMR spectroscopy: The bromine atoms in the dibromo derivative of the adduct with maleic anhydride are cis-oriented (endo-4-Br, endo-5-Br), whereas the bromine atoms have a trans configuration (endo-4-Br, exo-5-Br) in the dibromo derivative of the adduct with N-phenylmaleinimide.
Similar content being viewed by others
Literature cited
A. F. Oleinik, E. V. Adamskaya, K. Yu. Novitskii, N. P. Solov'eva, N. I. Fadeeva, I. N. Degtyareva, N. B. Lapaeva, and G. N. Pershin, Khim.-farm. Zh., No. 3, 49 (1980).
M. W. Lee and W. C. Herndon, J. Org. Chem., 43, 518 (1978).
S. Seltzer, J. Am. Chem. Soc., 87, 1534 (1965).
V. F. Bystrov, Usp. Khim., 12, 531 (1972).
I. S. Nikolaeva, T. V. Pushkina, G. N. Pershin, A. F. Oleinik, E. V. Adamskaya, and E. A. Golovanova, Khim.-farm. Zh., No. 8, 50 (1981).
J. Berson and R. Swidler, J. Am. Chem. Soc., 76, 4060 (1954).
F. A. Anet, Can. J. Chem., 39, 789 (1961).
S. Sternhell, Q. Rev. Chem. Soc., 23, 236 (1969).
A. Gordon and R. Ford, The Chemist's Companion, Wiley (1973).
Author information
Authors and Affiliations
Additional information
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 5, pp. 613–615, May, 1984.
Rights and permissions
About this article
Cite this article
Solov'eva, N.P., Sheinker, Y.N., Oleinik, A.F. et al. Investigation of the stereochemistry of adducts of aryloxyfurans with maleic acid derivatives by PMR spectroscopy. Chem Heterocycl Compd 20, 489–491 (1984). https://doi.org/10.1007/BF00514297
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00514297