Summary
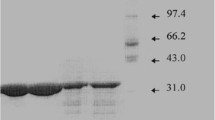
A procedure for purification of a serine alkaline proteinase from Streptomyces rimosus waste broth was developed. The procedure includes ultrafiltration, CM-Sephadex C-50 and CM-cellulose chromatography and gel filtration on Sephadex G-75. The recovery of electrophoretically pure enzyme was 25%. The enzyme is active on different protein and synthetic substrates at neutral and alkaline pHs but its activity on hemoglobin and bovine serum albumin, is optimal at pH 4.0–4.5. It has a molecular weight of 22,000 and a pI of 4.90. The enzyme is inhibited by DFP and PMSF but not by TPCK. Its substrate specificity, amino acid composition and some other properties were also determined.
Similar content being viewed by others
References
Andrews P (1965) The gel filtration behaviour of proteins related to their molecular weights over a wide range. Biochem J 96:595–606
Anson ML (1939) The estimation of pepsin, trypsin, papain and cathepsin with hemoglobin. J Gen Physiol 22:79–89
Awad WM, Soto AR, Siegel S, Shiba WE, Bernstrom GG, Ochoa MS (1972) The proteolytic enzymes of the K-1 strain Streptomyces griseus obtained from a commercial preparation (Pronase). J Biol Chem 247:4144–4155
Chauvet J, Dostal J, Acher R (1976) Isolation of a trypsin-like enzyme from Streptomyces paromomycinus (paromotrypsin) by affinity adsorption through Kunitz inhibitor-Sepharose. Int J Peptide Prot Res 8:45–55
Davis BJ (1964) Disc electrophoresis. 2. Method and application to human serum protein. Ann N.Y Acad Sci 121:404–427
Fremel VB, Shishkova EA (1968) Properties of proteolytic enzymes contained in the culture fluid of Actinomyces rimosus. Prikl Biokhim Mikrobiol 4:627–633
Gertler A, Trop M (1971) The elastase-like enzymes from Streptomyces griseus (Pronase), isolation and partial characterization. Eur J Biochem 19:90–96
Houmard J, Drapeau GR (1972) Staphilococcal protease. A proteolytic enzyme specific for glutamoyl bonds. Proc Natl Acad Sci USA 69:3506–3509
Kunitz MJ (1947) Crystalline soybean trypsin inhibitor. II. General properties. J Gen Physiol 30:291–310
Lowry OH, Rosenbrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193: 265–275
Morihara K (1974) Comparative Specificity of Microbial Proteinases. In: Meister A (ed) Advances in enzymology. John Wiley and Sons Inc, New York, pp 179–243
Nagatsu I, Nagatsu T, Yamamoto T, Glenner GS, Mehl JW (1970) Purification of aminopeptidase in human serum and degradation of angiotensin II by the purified enzyme. Biochim Biophys Acta 198:255–270
Narahashi Y, Yoda K (1977) J Biochem 81:587–597
Pokorny M, Vitale Lj, Turk V, Renko M, Žuvanić J (1979) Streptomyces rimosus extracellular proteases. 1. Characterization and evaluation of various crude preparations. Eur J Appl Microbiol Biotechnol 8:81–90
Pokorny M, Vitale Lj (1980) Enzymes as by-products during biosynthesis of antibiotics. In: Vitale Lj, Simeon V (eds) Industrial and clinical enzymology. Trends in enzymology. Pergamon Press, Oxford, pp 13–25
Radžabov LR, Shibnev VA, Rassulin YuA (1977) Esterase and peptidase activity of Actinomyces rimosus elastase. Khim Prir Soed 5:693–695
Rassulin YuA, Kaverzneva ED, Erkomaishvili GS (1972) Characteristics of the elastolytic complex from Actinomyces rimosus-rimoprotelin. Prikl Biokhim Mikrobiol 8:19–25
Rassulin YuA, Kaverzneva ED, Kobanova NK (1973a) Isolation and some Properties of a Trypsin-Like Enzyme from Actinomyces rimosus. In: Sadaukas PB (ed) Khimia Proteolitičeskih Fermentov. Akadmia Nauk, Vilnius, pp 100–101
Rassulin YuA, Kaverzneva ED, Erkomaishvili GS (1973b) Enzymatic activity of components of an elastase complex of Actinomyces rimosus. Biokhimia 38:727–729
Rassulin YuA, Radžabov LR, Kaverzneva ED, Shibnev VA, Erkomaishvili GS (1974) Isolation and properties of an elastolytic enzyme of Actinomyces rimosus. Izv Akad Nauk SSSR Ser Khim 3:687–693
Shotton DM (1970) Elastase. In: Collowick Sp, Kaplan NO (eds) Methods in enzymology, vol 19. Academic Press, New York, pp 113–140
Simpson RJ, Neuberger MR, Liu TY (1976) Complete amino acid analysis of proteins from a single hydrolyzate. J Biol Chem 251:1936–1940
Visser L, Blout ER (1972) The use of p-nitrophenyl N-tert-butyloxycarbonyl-l-alaninate as substrate for elastase. Biochim Biophys Acta 268:257–260
Walsh KA (1970) Trypsinogens and trypsins of various species. In: Collowick SP, Kaplan NO (eds) Methods in enzymology, vol 19. Academic Press, New York, pp 41–63
Weber K, Osborn M (1969) The reliability of molecular weight determination by dodecyl sulphate polyacrylamide gel electrophoresis. J Biol Chem 244:4406–4412
Yoshida E, Noda H (1965) Isolation and characterization of collagenases I and II from Chlostridium histolyticum. Biochim Biophys Acta 105:562–574
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Renko, M., Pokorny, M., Vitale, L. et al. Streptomyces rimosus extracellular proteases. European J. Appl. Microbiol. Biotechnol. 11, 166–171 (1981). https://doi.org/10.1007/BF00511256
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00511256