Summary
-
1.
In guinea-pig papillary muscle PCMBS (0.03–0.6 mmol/l) produced, after a transient negative inotropic phase, a pronounced positive inotropic effect. Finally a contracture developed, while the force of contraction declined continuously.
-
2.
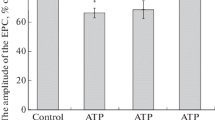
Endogenously released noradrenaline contributed to the time course but not to the maximum of the positive inotropic effect of PCMBS. The latter was equal to the maximal effect obtained by noradrenaline. For further evaluation of PCMBS effects, only muscles from animals pretreated with reserpine were used.
-
3.
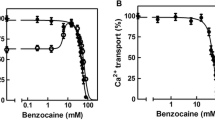
The increase of the concentration of PCMBS (from 0.03–0.6 mmol/l) did not significantly alter the magnitude of its negative or positive inotropic effects, but distinctly accelerated their development. When contraction force was plotted versus timex[PCMBS], the time curves of the development of the inotropic effects became superimposable. The similar dependence of negative as well as positive inotropic effects on the concentration of PCMBS suggested that the same type of receptor, probably sulfhydryl groups, is involved in the mediation of both effects.
-
4.
A mathematical analysis made evident that either the rate of permeation of PCMBS across some barrier far from equilibrium or the rate of binding of supramaximal concentrations of PCMBS to a receptor may be rate-limiting. Permeation is most likely the rate-limiting step, since the rapid inactivation of isolated sarcolemmal ATPase indicates a fast binding of PCMBS.
-
5.
The negative inotropic effects of PCMBS correlated with a shortening of both contraction time and plateau duration of the action potential. This made it probable that the negative inotropic effect and the shortening of action-potential duration were causally related.
-
6.
A reduction of [Na+]0 (from 140 to 70 mmol/l) or of stimulation frequency (from 1 to 0.004 Hz) or an increase of [K+]0 (from 2.4 to 9.6 mmol/l) delayed the appearance of the positive inotropic effect of PCMBS (t 50% of maximal effects was 4 min at 2.4 mmol/l K+ and 27 min at 9.6 mmol/l).
-
7.
Mg2++Na++K+- and Mg2+-dependent ATPases of sarcolemmal particles were inhibited by PCMBS (1–30 μmol/l).
-
8.
The similarities in the influence of variation of contraction frequency and of [Na+]0 or [K+]0 on the positive inotropic effects of PCMBS and of cardioactive glycosides and their common inhibitory action on membrane ATPase suggest that inhibition of this enzyme by the Hg-compound is the cause of its positive inotropic effect.
Similar content being viewed by others
References
Caprio A, Farah A (1967) The effect of ionic milieu on the response of rabbit cardiac muscle to ouabain. J Pharmacol Exp Ther 155:403–415
Ebner F, Reiter M (1977) The dependence on contraction frequency of the positive inotropic effect of dihydro-ouabain. Naunyn-Schmiedeberg's Arch Pharmacol 300:1–9
Ebner F, Reiter M (1979) The influence of (+)-propranolol on the inotropic effect of dihydro-ouabain in relation to stimulation frequency. Naunyn-Schmiedeberg's Arch Pharmacol 307:105–113
Ebner F, Bachmaier A, Reiter M (1979) The influence of K+ on the positive inotropic effect of dihydro-ouabain in relation to stimulation frequency and Ca2+. Naunyn-Schmiedeberg's Arch Pharmacol 308:R35
Fiske CH, Subbarow Y (1925) The colorimetric determination of phosphorus. J Biol Chem 66:375–400
From AHL, Probstfield JL (1971) p-Chloromercuribenzenesulfonic acid induced inotropism. Fed Proc 30:2441
Halbach S (1975) Effect of mercuric chloride on contractility and transmembrane potential of the guinea-pig myocardium. Naunyn-Schmiedeberg's Arch Pharmacol 289:137–148
Halbach S, Schönsteiner G (1979) Two different effects of p-chloromercuriphenylsulfonic acid (PCMBS) on contractility of guinea-pig myocardium. Naunyn-Schmiedeberg's Arch Pharmacol 308:R34
Honerjäger P, Reiter M (1975) The relation between effects of veratridine on action potential and contraction in mammalian ventricular myocardium. Naunyn-Schmiedeberg's Arch Pharmacol 289:1–28
Isenberg G (1977) Cardiac Purkinje fibres. [Ca2+] i controls steady-state potassium conductance. Pflügers Arch 371:71–76
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–275
Meech RW, Strumwasser F (1970) Intracellular calcium injection in aplysia nerve cells. Fed Proc 29:834
Reiter M (1963) Die Beziehung von Calcium und Natrium zur inotropen Glykosidwirkung. Naunyn-Schmiedeberg's Arch Pharmacol 245:487–499
Reiter M (1965) Die Untersuchung inotrop wirkender Substanzen. In: Festschrift 75 Jahre Hommel. Hommel, Zürich, pp 480–484
Reiter M (1972) Drugs and heart muscle. Ann Rev Pharmacol 12:111–124
Reiter M, Stickel FJ (1968) Der Einfluß der Kontraktionsfrequenz auf das Aktionspotential des Meerschweinchen-Papillarmuskels. Naunyn-Schmiedeberg's Arch Pharmacol 260:342–365
Reiter M, Strobl F (1972) Contraction frequency and concentration in the positive inotropic effect of a cardioactive steroid on ventricular muscle. J Pharmacol (Paris) 3:22
Reiter M, Stickel FJ, Weber S (1966) The influence of the extracellular potassium concentration on the glycoside effects upon contractile force and action potential duration of the guinea-pig papillary muscle. Experientia 22:665
Rothstein A (1970) Sulfhydryl groups in membrane structure and function. In: Bonner F, Kleinzeller A (eds) Current topics in membrane and transport. Academic Press, New York, pp 135–176
Sanyal PN, Saunders PR (1958) Relationship between cardiac rate and positive inotropic action of ouabain. J Pharmacol Exp Ther 122:499–503
Skou JC, Hilberg C (1965) The effect of sulphydryl-blocking reagents and of urea on the (Na++K+)-activated enzyme system. Biochim Biophys Acta 110:359–369
Tada M, Finney JO, Swartz MH, Katz AM (1972) Preparation and properties of plasma membranes from guinea pig hearts. J Mol Cell Cardiol 4:417–425
Temma K, Akera T, Ku DD, Brody TM (1978) Sodium pump inhibition by sulfhydryl inhibitors and myocardial contractility. Naunyn-Schmiedeberg's Arch Pharmacol 302:63–71
Weinryb I, Michel IM, Alicino JF, Hess SM (1971) Effects of barbiturate derivatives, sulfhydryl reagents, and oxidzing agents on the activity of adenylate cyclase from guinea pig heart and lung. Arch Biochem Biophys 146:591–596
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Halbach, S., Schönsteiner, G., Ebner, F. et al. The effects of p-chloromercuriphenylsulfonic acid (PCMBS) on force of contraction of mammalian myocardium and on ATP hydrolysis by sarcolemmal ATPase. Naunyn-Schmiedeberg's Arch. Pharmacol. 318, 121–129 (1981). https://doi.org/10.1007/BF00508836
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00508836