Abstract
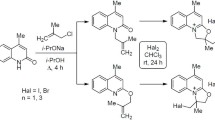
The methylation of 1-benzyl-Δ9,10-octahydro-4-quinolone with methyl iodide in the presence of lithium diethylamide in tetrahydrofuran is a regioselective electrophilic substitution reaction, and, depending on the reaction conditions, takes place in the 3 or 8 position of the quinolone system. Deuteration under the same conditions takes place only in the 3 position.
Similar content being viewed by others
Literature cited
G. V. Grishina, A. I. Vovk, and V. M. Potapov, Khim. Geterotsikl. Soedin., No. 11, 1565 (1979).
G. C. Levy and G. L. Nelson, Carbon-13 Nuclear Magnetic Resonance for Organic Chemists, Wiley, New York (1972).
V. A. Roznyatovskii, V. A. Chertkov, and Yu. K. Grishin, Zavod. Lab., 46, 805 (1980).
N. M. Sergeev, NMR Spectroscopy for Organic Chemists [in Russian], Izd. MGU, Moscow (1981), Chapter 4.
V. M. Potapov, G. V. Kiryushkina, G. N. Koval', and G. P. Tokmakov, Khim. Geterotsikl. Soedin., No. 4, 541 (1972).
L. Horii, C. Iwata, and G. Tamura, Chem. Pharm. Bull., 10, 940 (1962).
Author information
Authors and Affiliations
Additional information
Communication 1 from the series “Methylated cis-enamino ketones.”
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 6, pp. 780–784, June, 1982.
Rights and permissions
About this article
Cite this article
Grishina, G.V., Leshcheva, I.F., Sergeev, N.M. et al. Regioselective methylation of 1-benzyl-Δ9, 10-octahydro-4-quinolone. Chem Heterocycl Compd 18, 589–593 (1982). https://doi.org/10.1007/BF00506149
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00506149