Summary
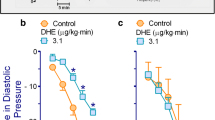
α-Methyldopa (10–100 mg/kg i.v.) produced a dose-dependent pupillary dilation in anaesthetized cats which was antagonized by subsequent administration of yohimbine hydrochloride (0.5 mg/kg i.v.). The peak effects were observed approximately 2–3h after injection. This α-methyldopa-induced mydriasis was present only when the parasympathetic innervation to the iris was intact. Prior treatment with yohimbine (0.5 mg/kg i.v.) 30 min before α-methyldopa also antagonized the mydriatic effect, whereas pretreatment with phenoxybenzamine (2.5 mg/kg i.v.) did not. In contrast, phenoxybenzamine, but not yohimbine, effectively antagonized the pupillary dilation produced by adrenaline (0.3–10.0 μg/kg i.v.). These results suggest that α-methyldopa produces mydriasis in the cat by means of CNS inhibition of tonic outflow from the oculomotor nucleus and that an α-adrenergic inhibitory mechanism may be involved. This conclusion is supported further by experiments in which direct measurements of ciliary nerve activity were made.
Similar content being viewed by others
References
Finch L, Haeusler G (1973) Further evidence for a central hypotensive action of α-methyldopa in both the rat and cat. Br J Pharmacol 47:217–228
Gherezghiher T, Koss MC (1979) Clonidine mydriasis in the rat. Eur J Pharmacol 57:263–266
Haeusler G (1973) Activation of the central pathway of the baroreceptor reflex, a possible mechanism of the hypotensive action of clonidine. Naunyn-Schmiedeberg's Arch Pharmacol 278:231–246
Haeusler G (1974) Clonidine-induced inhibition of sympathetic nerve activity: no indication for a central presynaptic or an indirect sympathomimetic mode of action. Naunyn-Schmiedeberg's Arch Pharmacol 286:97–111
Haeusler G (1977) Neuronal mechanisms influencing transmission in the baroreceptor reflec arc. In: De Jong W, Provoost AP, Shapiro AP (eds) Hypertension and brain mechanisms. Elsevier, Amsterdam, pp 95–109
Hamel EG, Jr, Kaelber WW (1961) Reserpine action on the central nervous system of the cat. Am J Physiol 200:195–200
Heise A, Kroneberg G (1973) Central nervous α-adrenergic receptors and the mode of action of α-methyldopa. Naunyn-Schmiedeberg's Arch Pharmacol 279:285–300
Henning M, van Zwieten PA (1968) Central hypotensive effect of α-methyldopa. J Pharm Pharmacol 20:409–417
Hoyer I, van Zwieten PA (1972) The central hypotensive action of amphetamine, ephedrine, phentermine, chlorphentermine and fenfluramine. J Pharm Pharmacol 24:452–458
Kobinger W (1978) Central α-adrenergic systems as targets for hypotensive drugs. Rev Physiol Biochem Pharmacol 81:39–100
Kobinger W, Pichler L (1978) Differentiation of drugs acting centrally upon the cardiovascular system by means of sympathetic and vagal responses. Clin Exp Hyperten 1:229–249
Koss MC (1979a) Topical clonidine produces mydriasis by a central nervous system action. Eur J Pharmacol 55:305–310
Koss MC (1979b) Clonidine mydriasis in the cat: further evidence for a CNS postsynaptic action. Naunyn-Schmiedeberg's Arch Pharmacol 309:235–239
Koss MC (1980) Studies on the mechanism of amphetamine mydriasis in the cat. J Pharmacol Exp Ther 213:49–53
Koss MC, Christensen HD (1979) Evidence for a central postsynaptic action of clonidine. Naunyn-Schmiedeberg's Arch Pharmacol 307:45–50
Koss MC, San LC (1976) Analysis of clonidine-induced mydriasis. Invest Ophthalmol Visual Sci 15:566–570
Nisida I, Okada H (1959) On the role of the pupillo-constrictory centers in the adrenalin-mydriasis and-miosis. Joa J Physiol 9:348–358
Schmitt H, Schmitt H (1970) Interactions between 2-(2,6-dichlorophenylamino)-2-imidazole hydrochloride (St 155, Catapresan) and α-adrenergic blocking drugs. Eur J Pharmacol 9:7–13
Schmitt H, Schmitt H, Fenard S (1971) Evidence for an α-sympathomimetic component in the effects of catapresan on vasomotor centres: antagonism by piperoxane. Eur J Pharmacol 14:98–100
Schmitt H, Schmitt H, Fenard S (1973) Action of α-adrenergic blocking drugs on the sympathetic centres and their interactions with the central sympatho-inhibitory effect of clonidine. Arzneim Forsch 23:40–45
Sharpe LG, Pickworth WB, Martin WR (1977) Actions of amphetamine and antagonists on pupil diameter in the chronic sympathectomized dog. Psychopharmacology 53:115–120
Sigg EB, Sigg TD (1967) Adrenergic modulation of central function. In: Garattini S, Dukes MNG (eds). Antidepressant drugs Exerpta Med, Amsterdam, pp 172–178
Torchiana ML, Lotti VJ, Clark CM, Stone CA (1973) Comparison of centrally mediated hypotensive action of methyldopa and DOPA in cats. Arch Int Pharmacodyn 205:103–113
Ury B, Gellhorn E (1939) Role of the sympathetic system in reflex dilatation of pupil. J Neurophysiol 2:268–275
Yonkman FF, Stilwell D, Jeremias R (1944) The adrenolytic and sympatholitic actions of yohimbine and ethyl yohimbine. J Pharmacol Exp Ther 81:111–115
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Koss, M.C. Methyldopa produces central inhibition of parasympathetic activity in the cat. Naunyn-Schmiedeberg's Arch. Pharmacol. 314, 135–139 (1980). https://doi.org/10.1007/BF00504529
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00504529